Product Images Fulvestrant
View Photos of Packaging, Labels & Appearance
- figure1 - fulvestrant injection 1
- figure9 - fulvestrant injection 10
- figure10 - fulvestrant injection 11
- figure11 - fulvestrant injection 12
- figure12 - fulvestrant injection 13
- figure13 - fulvestrant injection 14
- figure14 - fulvestrant injection 15
- Hikma syringe - fulvestrant injection 16
- Fulvestrant carton - fulvestrant injection 17
- figure2 - fulvestrant injection 2
- figure3 - fulvestrant injection 3
- figure4 - fulvestrant injection 4
- figure5 - fulvestrant injection 5
- structure - fulvestrant injection 6
- figure6 - fulvestrant injection 7
- figure7 - fulvestrant injection 8
- figure8 - fulvestrant injection 9
Product Label Images
The following 17 images provide visual information about the product associated with Fulvestrant NDC 0143-9022 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure9 - fulvestrant injection 10
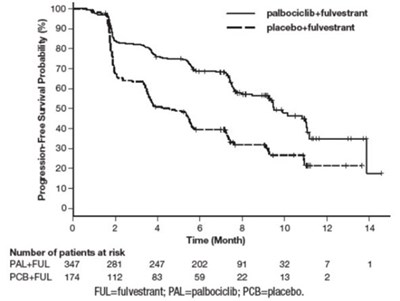
This is a chart showing the progression-free survival probability (%) over time (in months) for patients using different treatments (fulvestrant with either palbociclib or placebo). The numbers on the left show the percentage of survival probability and the numbers on the bottom show the time in months. The chart also shows the number of patients at risk for each treatment.*
figure10 - fulvestrant injection 11

This is a graph showing the comparison of Fulvestrant with and without Palbociclib and placebo. The graph compares the number of patients at risk over time (in months). The number of patients receiving Fulvestrant plus Palbociclib is noted as FULWPAL, Fulvestrant plus placebo is FULWPCB, and PCB represents the placebo group.*
figure11 - fulvestrant injection 12
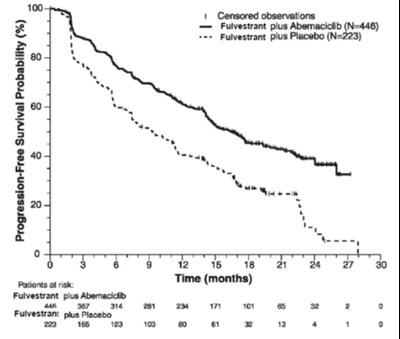
This appears to be a graph showing the Free Survival Probability of patients (represented by percentages) over a period of time (in months) for three different treatments. Two of the treatments are labeled as "Fulvestrant plus Placebo" and the third treatment is labeled as "Fulvestrant plus Abemacicib". There are censored observations on the graph as well.*
figure12 - fulvestrant injection 13
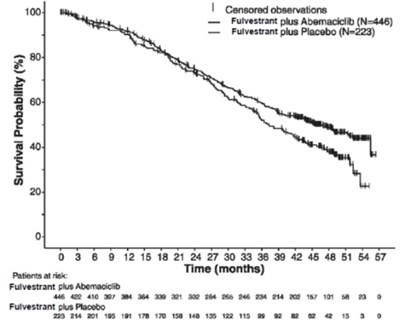
The text seems to be a table showing the results of a study that compares the effects of two different treatments (Fulvestrant plus Abemaciclib and Fulvestrant plus Placebo) over time (in months). Unfortunately, the process generated some errors, as there are some numbers and characters that do not seem to make sense or belong to the context. The text is not very readable, and it may be best to look for the original source of the data.*
Hikma syringe - fulvestrant injection 16

This is a description of Fulvestrant Injection which is a prescription medicine administered through intramuscular injection. The medicine comes in a single-dose pre-filled syringe of 250mg per 5mL (50mg per 1 mL). The NDC code for this medicine is 0143-9022-01. It should be noted that it is only meant for use as directed by a healthcare professional, and should be protected from light during storage. The rest of the text contains errors from and is not-useful.*
figure6 - fulvestrant injection 7
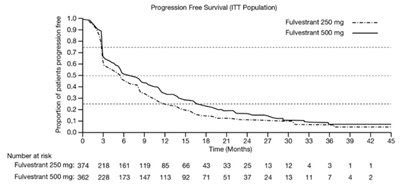
This is a table showing the progression-free survival of patients in two treatment groups - Fulvestrant 250mg and Fulvestrant 500mg. The table presents the proportion of patients who experienced a progression-free survival, along with the number of patients at risk in each group.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.