Product Images Dacarbazine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Dacarbazine NDC 0143-9245 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
vial - dacarbazine for injection usp 2

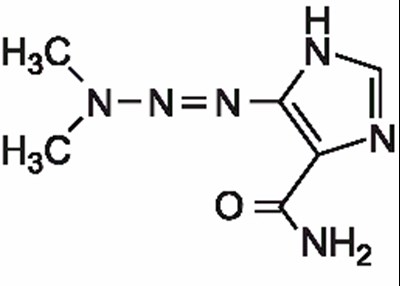
NDC 0143-9245-01 is Dacarbazine for Injection, USP with a dosage of 200 mg per citrate salt for intravenous use and is a cytotoi. The dosage recommendation is not available as the text is corrupted by and has missing information. The drug needs to be stored in a refrigerator at 2-8 degrees Celsius.*
CARTON - dacarbazine for injection usp 3

Dacarbazine for Injection, USP is a medication used for cancer treatment. It is not possible to determine more details from the scanned text.*
SERIALIZATION - dacarbazine for injection usp 4

This is a product identification code consisting of a Global Trade Item Number (GTIN), serial number (SN), expiration date (EXP), and lot number (LOT). Not available for further information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.