Product Images Dihydroergotamine Mesylate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Dihydroergotamine Mesylate NDC 0143-9273 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
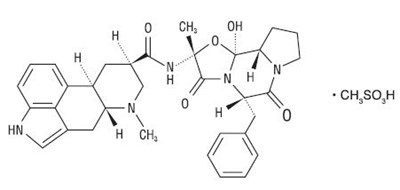
section D - dihydroergotamine mesylate injection usp 2
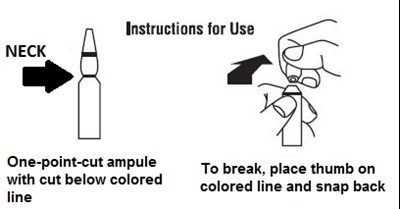
These are the instructions for use for a one-point-cut ampule that is to be used on the neck. There is a colored line below which the ampule should be cut. To break the ampule, place your thumb on the colored line and snap back.*
Container label - dihydroergotamine mesylate injection usp 3
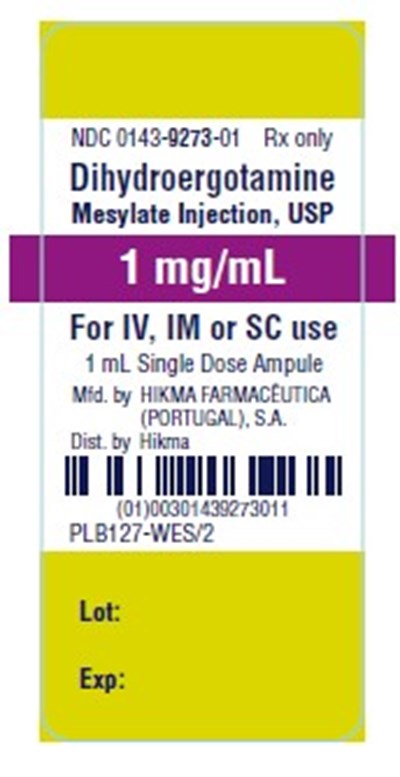
This is a description of a medication called Dihydroergotamine Mesyate injection, which is a solution containing 1mg/ml. It is intended for intravenous (IV), intramuscular (IM) or subcutaneous (SC) use and comes in a single-use ampule of 1 mL. The manufacturer is A Farmaceutica located in Portugal. The lot number is not available.*
dihydroergotamine mesylate injection usp 4
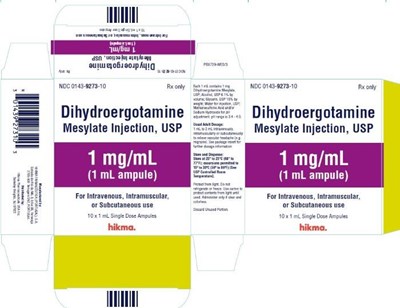
This is a description of a medication called Dihydroergotamine Mesylate Injection, USP. It comes in 1mg/mL ampules for intravenous, intramuscular or subcutaneous use. The medication is labeled with the code NDG 0143827310 Rxonly and NOC 0143827310 Rconly. The manufacturer appears to be Hikma and there are both 1 mL and single-dose ampules available.*
dihydroergotamine mesylate injection usp 5

This is a set of product identification codes. The GTIN (Global Trade Item Number) is 0301234567896, the SN (Serial Number) is 1234567890123, the EXP (Expiration Date) is MMMYYYY, and the LOT is ABCDE12345.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.