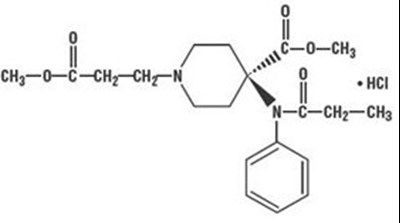
Product Images Remifentanil Hydrochloride
View Photos of Packaging, Labels & Appearance
- Image - 0bc4c167 59ce 43ad 9db8 281e4a166887 01
- Image - 0bc4c167 59ce 43ad 9db8 281e4a166887 02
- Image - 0bc4c167 59ce 43ad 9db8 281e4a166887 03
- 1 mg vial - 0bc4c167 59ce 43ad 9db8 281e4a166887 04
- 2mg vial - 0bc4c167 59ce 43ad 9db8 281e4a166887 05
- 5mg vial - 0bc4c167 59ce 43ad 9db8 281e4a166887 06
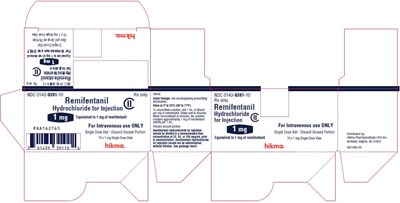
- 1 mg carton - 0bc4c167 59ce 43ad 9db8 281e4a166887 07
- 2 mg carton - 0bc4c167 59ce 43ad 9db8 281e4a166887 08
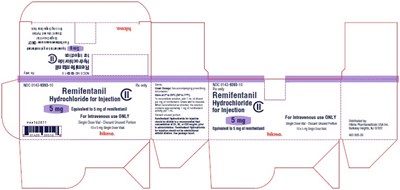
- 5 mg carton - 0bc4c167 59ce 43ad 9db8 281e4a166887 09
- serialization image - 0bc4c167 59ce 43ad 9db8 281e4a166887 10
Product Label Images
The following 10 images provide visual information about the product associated with Remifentanil Hydrochloride NDC 0143-9391 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - 0bc4c167 59ce 43ad 9db8 281e4a166887 02
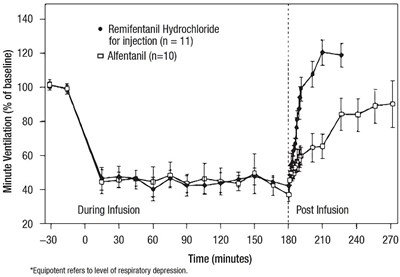
This is a graph showing the post-infusion effects of Remifentanil Hydrochloride and Alfentanil. The graph has a time scale (in minutes) on the horizontal axis and an equipotent level on the vertical axis. The equipotent level is a measure of respiratory depression caused by a particular dose of the drug. The graph seems to show that after an initial increase in equipotent level, Remifentanil Hydrochloride causes a significant decrease in respiratory depression compared to Alfentanil.*
Image - 0bc4c167 59ce 43ad 9db8 281e4a166887 03
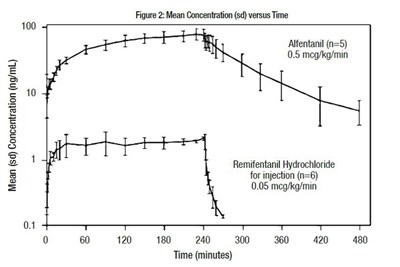
The text provides a graph with the mean concentration of Affentanil and Remifentanil Hydrochloride at different time intervals. The concentration is measured in nanograms per milliliter. The figure shows that Affentanil was administered to 5 individuals at a dose of 0.5 micrograms per kilogram per minute, while Remifentanil Hydrochloride was administered to 6 individuals at a dose of 0.05 micrograms per kilogram per minute. The graph displays the mean concentration with standard deviation over a period of 480 minutes (8 hours).*
1 mg vial - 0bc4c167 59ce 43ad 9db8 281e4a166887 04

Remifentanil is a prescription drug administered through injection or intravenous technique. It comes in single-dose vials that need to be discarded if unused. The drug is sterile and requires reconstitution and dilution before use, and the usual dosage should be referred to by checking the insert. The manufacturer distributing it is Hikma, and the product's NDC ID is 0143-9391-01.*
2mg vial - 0bc4c167 59ce 43ad 9db8 281e4a166887 05

This is a product description for a medication called Remifentanil Hydrochloride, which is for intravenous use only. The medication comes in a single dose vial and must be discarded if any portion remains unused. It is sterile, but must be reconstituted and diluted before use. The recommended dosage is not provided in the description and should be referenced in the product insert. The medication is distributed by Hikma and its National Drug Code (NDC) is 0143-9392-01.*
5mg vial - 0bc4c167 59ce 43ad 9db8 281e4a166887 06

Remifentanil is a prescription medication that comes in a single dose vial for intravenous use only. The vial contains 9 mg of Remifentanil Hydrochloride Il that must be reconstituted and diluted before use. Any unused portion should be discarded. The usual dosage information can be found in the insert. This medication is sterile and distributed by Hikma from Berkeley Heights in New Jersey. The product code is PAA162770 and the National Drug Code is 0143-9393-01.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.