Product Images Ketamine Hydrochloride
View Photos of Packaging, Labels & Appearance
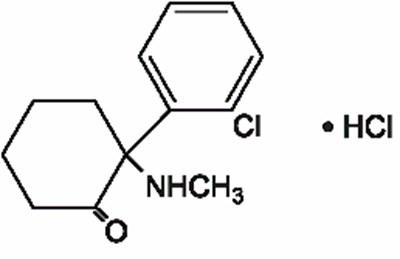
- structural formula - ketamine hydrochloride injection usp 1
- Ketamine Hydrochloride Injection, USP 50 mg/ml; 10 mL vial label - ketamine hydrochloride injection usp 2
- Ketamine Hydrochloride Injection, USP 100 mg/ml; 5 mL vial label - ketamine hydrochloride injection usp 3
- ketamine hydrochloride injection usp 4
- ketamine hydrochloride injection usp 5
- ketamine hydrochloride injection usp 6
Product Label Images
The following 6 images provide visual information about the product associated with Ketamine Hydrochloride NDC 0143-9509 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Ketamine Hydrochloride Injection, USP 50 mg/ml; 10 mL vial label - ketamine hydrochloride injection usp 2

Ketamine Hydrochloride Injection, USP 100 mg/ml; 5 mL vial label - ketamine hydrochloride injection usp 3

Ketamine is an injectable medication used for slow IV or IM use. It comes in a 5mL multiple dose vial, containing 500 mg of Ketamine Hydrochloride per 5 mL (100mg per ml). The solution should be diluted before IV use and is sterile. This medication is only available through a prescription (Rx Only) and the usual dosage should be determined by a physician. The solution contains benzethonium chloride as a preservative and should be protected from light, kept out of reach of children, and stored at 20° to 25°C (68° to 77°F). The packaging was manufactured by Hikma Farmaceltica (Portugal) and is distributed by Hiema.*
ketamine hydrochloride injection usp 4

This is a description of a medication identified by its National Drug Code (NDC) 0143-9508-01, which is only available with a prescription as it is labeled "Rxonly." The medication is sterile and is an injection of Ketamine. The injection is specified to be used for intramuscular administration at room temperature. The dosage strength and volume are not legible. The injection is contained in a 10 mL multiple-dose vial with benzethonium chloride solution added as a preservative. The medication is distributed by Hina in Berkeley Heights, J, with a lot number of PLB938-WES/2.*
ketamine hydrochloride injection usp 5

Ketamine HCI Injection is a prescription medication that comes in a 10mL multiple-dose vial. Each mL of the drug contains ketamine hydrochloride that is equivalent to 50mg of ketamine with a pH range of 3.5 to 5.5. It can be administered through the muscle or slowly through the vein. The solution is sterile and must be protected from light. The manufacturer is HIKMA FARMACELTICA while the distributor is Iema located in Berkeley Heights. The usual dosage should be seen in the package insert.*
ketamine hydrochloride injection usp 6

This is a product identification information including GTIN (Global Trade Item Number), SN (Serial Number), EXP (Expiration Date), and LOT (Lot Number). It can be used to track and trace product information for inventory and quality control purposes.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.