Product Images Metoprolol Tartrate
View Photos of Packaging, Labels & Appearance
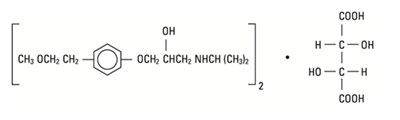
- chemical structure - metoprolol tartrate injection 1
- NDC 0143-9873-10 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL (1 mg/mL) FOR IV USE ONLY Rx ONLY 5 mL Single Dose Vial Each 5 mL vial contains: 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. USUAL DOSAGE: See package insert. DISCARD UNUSED PORTION. Protect from light. Do not freeze. - metoprolol tartrate injection 2
- NDC 0143-9873-10 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL Rx only (1 mg/mL) DISCARD UNUSED PORTION. FOR IV USE ONLY 10 x 5 mL Single Dose Vials Each 5 mL contains 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. USUAL DOSAGE: See package insert. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Protect from light. Do not freeze. Retain in carton until time of use. - metoprolol tartrate injection 3
- NDC 0143-9873-25 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL Rx only (1 mg/mL) DISCARD UNUSED PORTION. FOR IV USE ONLY 25 x 5 mL Single Dose Vials Each 5 mL contains 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. USUAL DOSAGE: See package insert. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Protect from light. Do not freeze. Retain in carton until time of use. - metoprolol tartrate injection 4
- NDC 0143-9660-01 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL (1 mg/mL) FOR IV USE ONLY Rx ONLY 5 mL Single Dose Vial Each 5 mL vial contains: 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. USUAL DOSAGE: See package insert. DISCARD UNUSED PORTION. Protect from light. Do not freeze. - metoprolol tartrate injection 5
- NDC 0143-9660-10 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL Rx only (1 mg/mL) DISCARD UNUSED PORTION. FOR IV USE ONLY 10 x 5 mL Single Dose Vials Each 5 mL contains 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. USUAL DOSAGE: See package insert. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Protect from light. Do not freeze. Retain in carton until time of use. - metoprolol tartrate injection 6
- metoprolol tartrate injection 7
Product Label Images
The following 7 images provide visual information about the product associated with Metoprolol Tartrate NDC 0143-9660 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
NDC 0143-9873-10 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL (1 mg/mL) FOR IV USE ONLY Rx ONLY 5 mL Single Dose Vial Each 5 mL vial contains: 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. USUAL DOSAGE: See package insert. DISCARD UNUSED PORTION. Protect from light. Do not freeze. - metoprolol tartrate injection 2
![metoprolol-tartrate-injection-2.jpg NDC 0143-9873-10 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL (1 mg/mL) FOR IV USE ONLY Rx ONLY 5 mL Single Dose Vial Each 5 mL vial contains: 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. USUAL DOSAGE: See package insert. DISCARD UNUSED PORTION. Protect from light. Do not freeze. - metoprolol tartrate injection 2](https://ndclist.com/assets/spl/images/c7ec36bb-bc6f-42de-9974-a50ac13e9410/400x-metoprolol-tartrate-injection-2.jpg)
This is a description for a medication called Metoprolol Tartrate Injection, USP. It comes in a single-dose vial with 5mg per 5ml (1mg/ml) for intravenous use only.*
NDC 0143-9873-10 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL Rx only (1 mg/mL) DISCARD UNUSED PORTION. FOR IV USE ONLY 10 x 5 mL Single Dose Vials Each 5 mL contains 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. USUAL DOSAGE: See package insert. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Protect from light. Do not freeze. Retain in carton until time of use. - metoprolol tartrate injection 3
![metoprolol-tartrate-injection-3.jpg NDC 0143-9873-10 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL Rx only (1 mg/mL) DISCARD UNUSED PORTION. FOR IV USE ONLY 10 x 5 mL Single Dose Vials Each 5 mL contains 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. USUAL DOSAGE: See package insert. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Protect from light. Do not freeze. Retain in carton until time of use. - metoprolol tartrate injection 3](https://ndclist.com/assets/spl/images/c7ec36bb-bc6f-42de-9974-a50ac13e9410/400x-metoprolol-tartrate-injection-3.jpg)
Metoprolol Tartrate Injection is a medication that is available in a 5 mg per 5 ml (1 mg/mL) strength in 10 x 5 mL single dose vials. It should be used intravenously only, and any unused portion should be discarded. The usual dosage should be determined by a healthcare professional, and the medication should be stored at 20°C to 25°C. The medication should be protected from light and should not be frozen. This product is distributed by Hika from Berley Heights.*
NDC 0143-9873-25 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL Rx only (1 mg/mL) DISCARD UNUSED PORTION. FOR IV USE ONLY 25 x 5 mL Single Dose Vials Each 5 mL contains 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. USUAL DOSAGE: See package insert. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Protect from light. Do not freeze. Retain in carton until time of use. - metoprolol tartrate injection 4
![metoprolol-tartrate-injection-4.jpg NDC 0143-9873-25 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL Rx only (1 mg/mL) DISCARD UNUSED PORTION. FOR IV USE ONLY 25 x 5 mL Single Dose Vials Each 5 mL contains 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. USUAL DOSAGE: See package insert. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Protect from light. Do not freeze. Retain in carton until time of use. - metoprolol tartrate injection 4](https://ndclist.com/assets/spl/images/c7ec36bb-bc6f-42de-9974-a50ac13e9410/400x-metoprolol-tartrate-injection-4.jpg)
This is a product description for PLEGSO-WES/3 Metoprolol Tartrate Injection, USP used for intravenous purposes. It comes in 25 single-dose vials each containing 5 mL of sterile solution with a dosage of 5 mg Metoprolol Tartate, USP, 45 mg sodium chloride, and water for injection, USP. The product should be stored at a temperature of 20° to 25°C (68° to 77°F), protected from light, and not frozen. It is distributed by Hikma in Berkeley Heights, NJ.*
NDC 0143-9660-01 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL (1 mg/mL) FOR IV USE ONLY Rx ONLY 5 mL Single Dose Vial Each 5 mL vial contains: 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. USUAL DOSAGE: See package insert. DISCARD UNUSED PORTION. Protect from light. Do not freeze. - metoprolol tartrate injection 5
![metoprolol-tartrate-injection-5.jpg NDC 0143-9660-01 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL (1 mg/mL) FOR IV USE ONLY Rx ONLY 5 mL Single Dose Vial Each 5 mL vial contains: 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. USUAL DOSAGE: See package insert. DISCARD UNUSED PORTION. Protect from light. Do not freeze. - metoprolol tartrate injection 5](https://ndclist.com/assets/spl/images/c7ec36bb-bc6f-42de-9974-a50ac13e9410/400x-metoprolol-tartrate-injection-5.jpg)
This is a product description for Metoprolol Tartrate Injection, which is a 5mg per 5mL (1mg/mL) solution used for intravenous use only. It comes in a single-dose vial of 5L. The manufacturer is Hauia Armaceuticr (Portugal), SA and the distributor is by Ik Pramcasscls USH Inc. The product is intended for usage after being directed by physicians and should be protected from light. No additional information was present in the text.*
NDC 0143-9660-10 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL Rx only (1 mg/mL) DISCARD UNUSED PORTION. FOR IV USE ONLY 10 x 5 mL Single Dose Vials Each 5 mL contains 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. USUAL DOSAGE: See package insert. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Protect from light. Do not freeze. Retain in carton until time of use. - metoprolol tartrate injection 6
![metoprolol-tartrate-injection-6.jpg NDC 0143-9660-10 METOPROLOL TARTRATE INJECTION, USP 5 mg/5 mL Rx only (1 mg/mL) DISCARD UNUSED PORTION. FOR IV USE ONLY 10 x 5 mL Single Dose Vials Each 5 mL contains 5 mg Metoprolol Tartrate, USP, 45 mg sodium chloride, Water for Injection, USP. USUAL DOSAGE: See package insert. Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature]. Protect from light. Do not freeze. Retain in carton until time of use. - metoprolol tartrate injection 6](https://ndclist.com/assets/spl/images/c7ec36bb-bc6f-42de-9974-a50ac13e9410/400x-metoprolol-tartrate-injection-6.jpg)
This is the product description of PLE788-WES/3, an injectable solution containing Metoprolol Tartrate, USP and Netoprolol Tartate, USP as active ingredients. The usual dosage and other information can be found in the package insert. The product comes in 10 x 5 mL Single Dose Vials from Hikma.*
metoprolol tartrate injection 7

This appears to be product information containing a GTIN code, serial number, expiration date, and lot number. The product can be identified using the GTIN code and the serial number, while the expiration date and lot number provide additional information about the product's manufacturing and expire date.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.