Product Images Betamethasone Dipropionate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Betamethasone Dipropionate NDC 0168-0268 by E. Fougera & Co. A Division Of Fougera Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 15 g Tube Box - 73e74ab9 6871 4784 bd46 5f29a3b89ad4 02

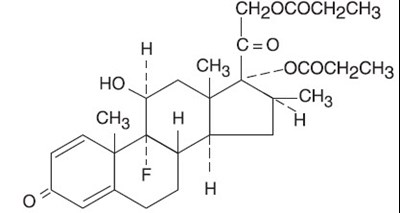
This is a product description for Fougera Betamethasone Dipropionate Ointment USP, an augmented ointment containing 0.05% Betamethasone Dipropionate, to be used topically for skin conditions. This product has a usual dosage of applying a thin film of ointment to the affected area, up to three or four times a day without covering it. Each gram contains 0.1543 g of USP Betamethasone Dipropionate, but it is not recommended for oral, ophthalmic, or intravaginal use. The cream comes in 15-gram tubes and should be stored at temperatures up to 25°C (77°F), should not be punctured or used if sealing is already damaged. The product should be kept out of the reach of children. There is a lot number and expiration date printed on the packaging.*
PRINCIPAL DISPLAY PANEL - 15 g Tube Box - 73e74ab9 6871 4784 bd46 5f29a3b89ad4 03

This is a topical ointment containing Betamethasone Dipropionate, used for skin conditions. It should not be used orally, in the eyes, or internally. The product is to be kept out of the reach of children and should not be used on children without consulting a physician. The strength of the ointment is 0.05%. The tube should not contain more than 50 grams, and a puncture seal ensures the product remains sterile. The manufacturers are Fougera Pharmaceuticals.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.