Product Images Rebinyn
View Photos of Packaging, Labels & Appearance
- Overview of REBINYN Package - image 01
- Symbol - image 02
- Vial Adapter - image 04
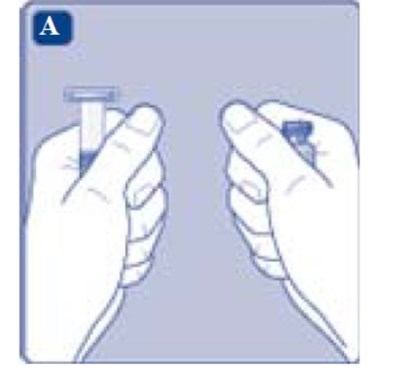
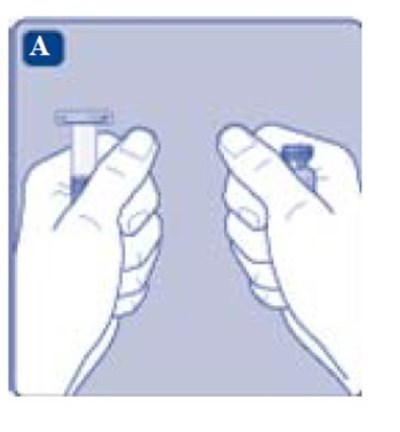
- Fig. A - image 05
- Fig. B - image 06
- Fig. C - image 07
- Fig. D - image 08
- Fig. E - image 09
- Fig. F - image 10
- FIg. G - image 11
- Fig. H - image 12
- Fig. I - image 13
- Fig. J - image 14
- Fig. K - image 15
- Pre-filled syringe with histidine diluent - image 16
- Plunger rod - image 17
- Fig. A - image 18
- Fig. B - image 19
- Fig. C - image 20
- Fig. D - image 21
- Fig. E - image 22
- Fig. F - image 23
- Fig. G - image 24
- Fig. H - image 25
- Fig. I - image 26
- Fig. J - image 27
- Symbol - image 28
- Fig. K - image 29
- Fig. L - image 30
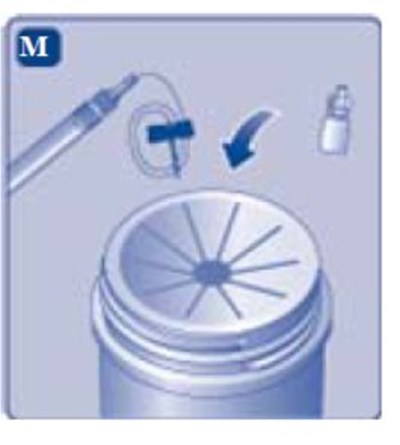
- Fig. M - image 31
- 1000 IU Carton - image 32
- 2000 IU Carton - image 33
- Histidine Solution - image 34
- 500 IU Carton - image 35
- 500 IU Vial - image 36
- Vial with Rebinyn Powder - image 37
- image 38
- 1000 IU Vial - image 39
Product Label Images
The following 38 images provide visual information about the product associated with Rebinyn NDC 0169-7902 by Novo Nordisk, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Vial Adapter - image 04

This text appears to be describing different components of a medical instrument or device. There is a vial adapter, a protective cap, a spike protective, and some kind of protective paper. Without additional context or visual aid, it is unclear what specific purpose these components serve or how they are used.*
Pre-filled syringe with histidine diluent - image 16

This appears to be a description of a pre-filled syringe that includes histidine diluent, a rubber plunger, a syringe tip, and a syringe cap. There is also a scale included.*
Plunger rod - image 17

The text describes a tool or object called a "plunger rod" that has a wide top end with threads.*
1000 IU Carton - image 32

This is a description of recombinant GlycoPEGylated Coagulation Factor IX medication, marketed under the name REBINYN and used to treat certain blood clotting disorders.*
2000 IU Carton - image 33

This is a product label for REBINYN®, which is a medication used to treat blood clotting disorders caused by low levels of Factor IX. REBINYN® is a recombinant, GlycoPEGylated version of Coagulation Factor IX. The medication comes in a 1000 U dose.*
Histidine Solution - image 34

This appears to be a product label for a medication. It includes the name of the medication "Coagulation Factor X Recombinan" and instructions on its storage conditions. There is also a warning to not freeze the medication. The text is not very clear and contains some misspelled words, making it difficult to fully understand.*
500 IU Carton - image 35

REBINYN® 2000 1U Range and REBINYN® 2000 10ss are two different products that contain coagulation factors. REBINYN® 2000 1U Range contains Factor IX (Recombinant), GlycoPEGylated, while REBINYN® 2000 10ss contains Factor X Mecambinam GyeoPecyited.*
500 IU Vial - image 36

This is a product label for a medication called REBINYN® Ll which is a recombinant coagulation factor IX that has been GiycoPEGylated. The medication is stored in a refrigerator with a certain amount of histidine and should not be frozen. The label contains product information including the product code, storage instructions, and pharmaceutical company details. The expiry and lot number are not readable.*
Vial with Rebinyn Powder - image 37

This is a description of a vial containing Rebinyn® powder. The vial has a plastic cap and a rubber stopper, which is located under the plastic cap.*
image 38

Product Name: REBINYN(R) 2000 1U Range. It contains Coagulation Factor X (Recombinant). NDC number is 01697922 11. No other useful information is available in the given text.*
1000 IU Vial - image 39

This is a product label for a 10 mM solution of Histidine used for the reconstitution of REBINYN. The NDC number, storage instructions, and a single-use only warning are given. There is also lot and expiration date information for the product.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.