Product Images Flovent Diskus
View Photos of Packaging, Labels & Appearance
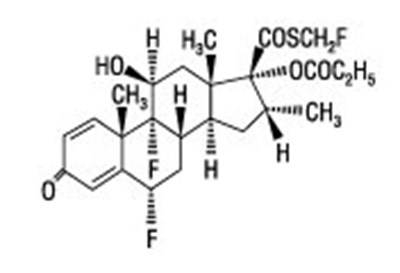
- Chemical structure - flovent diskus spl graphic 01
- Figure 1. A 12-Week Clinical Trial Evaluating FLOVENT DISKUS 100 mcg Twice Daily in Adults and Adolescents Receiving Bronchodilators Alone - flovent diskus spl graphic 02
- Figure 2. A 12-Week Clinical Trial Evaluating FLOVENT DISKUS 100 mcg Twice Daily in Adults and Adolescents Receiving Inhaled Corticosteroids - flovent diskus spl graphic 03
- Figure 3. A 12-Week Clinical Trial Evaluating FLOVENT DISKUS 250 mcg Twice Daily in Adults and Adolescents Receiving Inhaled Corticosteroids or Bronchodilators Alone - flovent diskus spl graphic 04
- Figure 4. A 12-Week Clinical Trial Evaluating FLOVENT DISKUS 500 mcg Twice Daily in Adults and Adolescents Receiving Inhaled Corticosteroids or Bronchodilators Alone - flovent diskus spl graphic 05
- Figure A - flovent diskus spl graphic 06
- Figure B - flovent diskus spl graphic 07
- Figure C - flovent diskus spl graphic 08
- Figure D - flovent diskus spl graphic 09
- Figure E - flovent diskus spl graphic 10
- Figure F - flovent diskus spl graphic 11
- Figure G - flovent diskus spl graphic 12
- Figure H - flovent diskus spl graphic 13
- Flovent Diskus 50 mcg 60 dose carton - flovent diskus spl graphic 14
- Flovent Diskus 100 mcg 60 dose carton - flovent diskus spl graphic 15
- Flovent Diskus 250 mcg 60 dose carton - flovent diskus spl graphic 16
Product Label Images
The following 16 images provide visual information about the product associated with Flovent Diskus NDC 0173-0601 by Glaxosmithkline Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. A 12-Week Clinical Trial Evaluating FLOVENT DISKUS 100 mcg Twice Daily in Adults and Adolescents Receiving Bronchodilators Alone - flovent diskus spl graphic 02
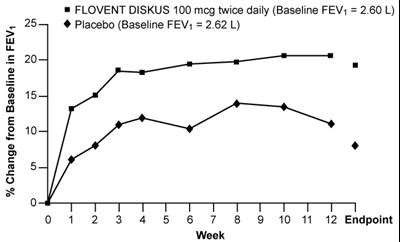
This is a comparison of the change in FEV1 (Forced Expiratory Volume in one second) from baseline between FLOVENT DISKUS 100 mcg taken twice a day and placebo in a clinical trial that lasted for 12 weeks. The baseline FEV1 was 2.60 L for FLOVENT DISKUS and 2.62 L for placebo. The data is presented graphically with the time points from 1 to 12 weeks along the horizontal axis and the change in FEV1 in percentages along the vertical axis.*
Figure 2. A 12-Week Clinical Trial Evaluating FLOVENT DISKUS 100 mcg Twice Daily in Adults and Adolescents Receiving Inhaled Corticosteroids - flovent diskus spl graphic 03
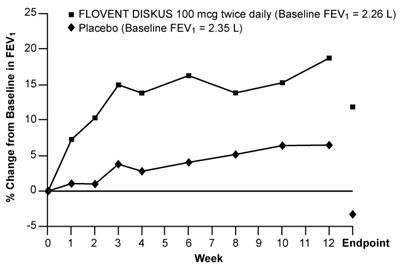
This is a chart showing the % change from baseline in FEV (Forced Expiratory Volume) for patients taking FLOVENT DISKUS 100 mcg twice daily (baseline FEV = 2.26 L) and those taking a placebo (baseline FEV = 2.35 L) over a period of 12 weeks.*
Figure 3. A 12-Week Clinical Trial Evaluating FLOVENT DISKUS 250 mcg Twice Daily in Adults and Adolescents Receiving Inhaled Corticosteroids or Bronchodilators Alone - flovent diskus spl graphic 04
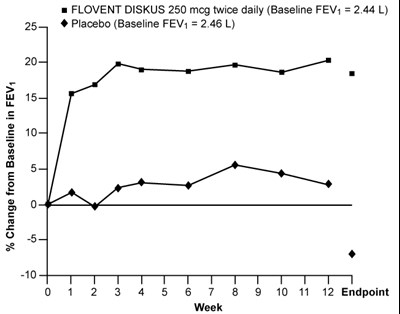
This is a clinical study comparison of administering FLOVENT DISKUS with a placebo on patients twice a day. The study tracked the change in FEV1 levels from baseline. Unfortunately, there is no data on the plotted graph to make any conclusions.*
Figure 4. A 12-Week Clinical Trial Evaluating FLOVENT DISKUS 500 mcg Twice Daily in Adults and Adolescents Receiving Inhaled Corticosteroids or Bronchodilators Alone - flovent diskus spl graphic 05
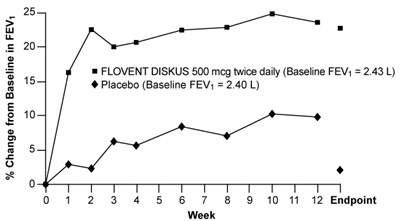
The text describes a chart or graph showing the percentage change from baseline in FEV (forced expiratory volume) for two different treatments - FLOVENT DISKUS 500 mcg twice daily and placebo. The graph shows a time frame of 12 endpoints, with each endpoint likely representing a measurement taken at a different time. The baseline FEV for FLOVENT DISKUS was 2.43 L and the baseline FEV for the placebo was 240 L.*
Flovent Diskus 50 mcg 60 dose carton - flovent diskus spl graphic 14
Flovent Diskus is a medication used for oral inhalation only, containing 50 mcg of fluticasone propionate with lactose in each blister. It comes in a diskus inhalation device with one foil strip of 60 blisters. Dispense with the enclosed Patient Information leaflet and refer to the prescribing information for dosage instructions.*
Flovent Diskus 100 mcg 60 dose carton - flovent diskus spl graphic 15

Flovent Diskus is an inhalation powder medication containing 100 mcg of fluticasone propionate with lactose per blister. It is intended for oral inhalation only and is dispensed with an enclosed patient information leaflet. The medication is available in a 1 Diskus Inhalation Device, which includes 1 foil strip of 60 blisters. Dosage information can be found in the prescribing information.*
Flovent Diskus 250 mcg 60 dose carton - flovent diskus spl graphic 16
Flovent Diskus is a powder medication for oral inhalation only that contains fluticasone propionate in 250 mcg doses in each blister, together with lactose. The medication comes in a Diskus Inhalation Device that contains one foil strip of 60 blisters. The patient information leaflet should be read before use. The prescribing information should be followed for dosage instructions.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.