Product Images Advair Diskus
View Photos of Packaging, Labels & Appearance
- Fluticasone propionate chemical structure - advair diskus spl graphic 01
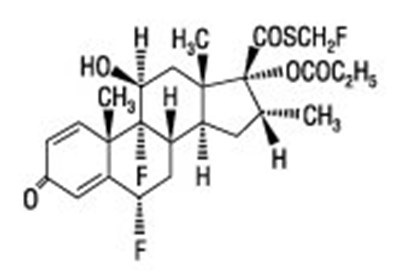
- Salmeterol xinafoate chemical structure - advair diskus spl graphic 02
- Figure 1. Mean Percent Change from Baseline in FEV1 in Subjects with Asthma Previously Treated with Either Inhaled Corticosteroids or Salmeterol (Trial 1) - advair diskus spl graphic 03
- S:\Project Files\A\Advair Diskus NDA 021077\Figures\advair-diskus-spl-graphic-04.jpg - advair diskus spl graphic 04
- Figure 3. Percent Change in Serial 12-Hour FEV1 in Subjects with Asthma Previously Using Either Inhaled Corticosteroids or Salmeterol (Trial 1) - advair diskus spl graphic 05
- Figure 4. Predose FEV1: Mean Percent Change from Baseline in Subjects with Chronic Obstructive Pulmonary Disease - advair diskus spl graphic 06
- Figure 5. Two-Hour Postdose FEV1: Mean Percent Changes from Baseline over Time in Subjects with Chronic Obstructive Pulmonary Disease - advair diskus spl graphic 07
- Figure A - advair diskus spl graphic 08
- Figure B - advair diskus spl graphic 09
- Figure C - advair diskus spl graphic 10
- Figure D - advair diskus spl graphic 11
- Figure E - advair diskus spl graphic 12
- Figure F - advair diskus spl graphic 13
- Figure G - advair diskus spl graphic 14
- Figure H - advair diskus spl graphic 15
- Advair Diskus 100mcg-50mcg carton - advair diskus spl graphic 16
- Advair Diskus 250mcg-50mcg carton - advair diskus spl graphic 17
- Advair Diskus 500mcg-50mcg carton - advair diskus spl graphic 18
Product Label Images
The following 18 images provide visual information about the product associated with Advair Diskus NDC 0173-0696 by Glaxosmithkline Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. Mean Percent Change from Baseline in FEV1 in Subjects with Asthma Previously Treated with Either Inhaled Corticosteroids or Salmeterol (Trial 1) - advair diskus spl graphic 03

S:\Project Files\A\Advair Diskus NDA 021077\Figures\advair-diskus-spl-graphic-04.jpg - advair diskus spl graphic 04

Figure 3. Percent Change in Serial 12-Hour FEV1 in Subjects with Asthma Previously Using Either Inhaled Corticosteroids or Salmeterol (Trial 1) - advair diskus spl graphic 05

Figure 4. Predose FEV1: Mean Percent Change from Baseline in Subjects with Chronic Obstructive Pulmonary Disease - advair diskus spl graphic 06

This appears to be a chart showing the results of a study on the effectiveness of ADVAIR DISKUS 250/50 and Salmeterol as compared to a placebo, based on the FEV1 (Forced Expiratory Volume in 1 second) of the participants. The chart shows the FEV1 measurements at various time intervals over the course of the study.*
Figure 5. Two-Hour Postdose FEV1: Mean Percent Changes from Baseline over Time in Subjects with Chronic Obstructive Pulmonary Disease - advair diskus spl graphic 07

This appears to be a table of data related to the performance of different inhalers. The inhalers compared are ADVAIR DISKUS 25050, Futcasons ropinals 250, and Placebo. The table provides the baseline FEV (forced expiratory volume) for each inhaler and displays the measurement of FEV again after a period of time has passed. The data is displayed in a chart or table format.*
Figure A - advair diskus spl graphic 08

This text provides a list of three separate elements likely related to a product or object. The first is an "Outer Case," which may refer to a protective cover or shell for some item. The second is a "Mouthpiece," suggesting that the item may be something that is held or inserted into the mouth, like a musical instrument or medical device. The third element is a "Thumb Grip," which potentially indicates a feature of the item that aids in gripping or maneuvering it with one's thumb. Without further context, it is difficult to determine the exact nature of the item, but it appears to be a physical object with these three distinct components.*
Advair Diskus 100mcg-50mcg carton - advair diskus spl graphic 16

This is a description for Advair IS0, an inhalation powder containing fluticasone propionate and salmeterol for oral use only. Each blister has 100mcg of fluticasone propionate and 72.5mcg of salmeterol xinafoate, equivalent to 50mcg of salmeterol base, along with lactose. This Advair IS0 package includes one Diskus inhalation device with one foil strip of 60 blisters.*
Advair Diskus 250mcg-50mcg carton - advair diskus spl graphic 17

This is a description of a medication called "ADVAIR DSKS" that comes in the form of an inhalation powder containing fluticasone propionate and salmeterol xinafoate. Each blister contains 250 mcg of fluticasone propionate and 72.5 mcg of salmeterol xinafoate, equivalent to 50 mcg of salmeterol base, with lactose. It should be used for oral inhalation only and requires a DISKUS inhalation device, which contains one foil strip of 60 blisters.*
Advair Diskus 500mcg-50mcg carton - advair diskus spl graphic 18

This is a medication called Advair Diskus, which is used for oral inhalation. Each blister contains 500mcg of fluticasone propionate and 72.5mcg of salmeterol. It comes in a device called DISKUS that contains 1 foil strip of 60 blisters.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.