Product Images Advair Hfa
View Photos of Packaging, Labels & Appearance
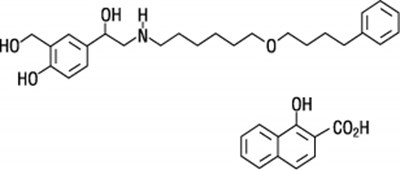
- Fluticasone propionate chemical structure - advairhfa spl graphic 01
- Salmeterol xinafoate chemical structure - advairhfa spl graphic 02
- Figure 1. Mean Percent Change from Baseline in FEV1 in Subjects Previously Treated with Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - advairhfa spl graphic 03
- Figure 2. Mean Percent Change from Baseline in Morning Peak Expiratory Flow in Subjects Previously Treated with Inhaled Corticosteroids (Trial 4) - advairhfa spl graphic 04
- Figure 3. Percent Change in Serial 12-Hour FEV1 in Subjects Previously Using Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - advairhfa spl graphic 05
- Figure 4. Percent Change in Serial 12-Hour FEV1 in Subjects Previously Using Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - advairhfa spl graphic 06
- Figure A - advairhfa spl graphic 07
- Figure B - advairhfa spl graphic 08
- Figure C - advairhfa spl graphic 09
- Figure D - advairhfa spl graphic 10
- Figure E - advairhfa spl graphic 11
- Figure F - advairhfa spl graphic 12
- Figure G - advairhfa spl graphic 13
- Figure H - advairhfa spl graphic 14
- Figure I - advairhfa spl graphic 15
- Advair HFA 45mcg 120 dose carton - advairhfa spl graphic 16
- Advair HFA 115mcg 120 dose carton - advairhfa spl graphic 17
- Advair HFA 230mcg 120 dose carton - advairhfa spl graphic 18
- advairhfa spl graphic 19
Product Label Images
The following 19 images provide visual information about the product associated with Advair Hfa NDC 0173-0717 by Glaxosmithkline Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. Mean Percent Change from Baseline in FEV1 in Subjects Previously Treated with Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - advairhfa spl graphic 03

This is a prescription for two inhalations of Advair HFA, which contains 45 micrograms/21 meg of medication, to be taken twice daily. The second line appears to be a different medication called Fluticasone Propionate Inhalation Aerosol at 4 micrograms per two inhalations, but the context of its use and frequency are not clear. There are also some scattered characters and symbols that do not provide additional relevant information.*
Figure 2. Mean Percent Change from Baseline in Morning Peak Expiratory Flow in Subjects Previously Treated with Inhaled Corticosteroids (Trial 4) - advairhfa spl graphic 04

Figure 3. Percent Change in Serial 12-Hour FEV1 in Subjects Previously Using Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - advairhfa spl graphic 05

This is a report on a study comparing the change in FEV (forced expiratory volume) for two groups of subjects who were treated with different inhalation treatments. One group received Advair HFA 45 mog/21 meg in two inhalations twice daily (n = 92), while the other group received fluticasone propionate inhalation aerosol 44 meg in two inhalations twice daily (1= 89) and salmeterol inhalation aerosol 21 meg in two inhalations twice daily. The report provides data in the form of a line graph that displays the change in FEV over time from Day 1 to the baseline.*
Figure 4. Percent Change in Serial 12-Hour FEV1 in Subjects Previously Using Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - advairhfa spl graphic 06

This text provides information on a study that compared the effectiveness of different inhalation aerosols for treating a condition that affects breathing. Specifically, it tested the efficacy of two doses of Advair HFA, a combination of fluticasone propionate and salmeterol, against a single-dose of fluticasone propionate, salmeterol, and placebo. The study analyzed the percentage change in forced expiratory volume (FEV) in one second, recorded at different times during the treatment, relative to the baseline FEV value on day one. The study lasted for twelve weeks.*
Figure A - advairhfa spl graphic 07

This text describes a metal canister with a purple plastic actuator and mouthpiece. It also includes a counter and a light purple cap.*
Figure E - advairhfa spl graphic 11

This text seems to be describing a certain position of a mouthpiece. There is a mention of the color "purple" and a "light" with a purple cap. However, there is not enough context to determine the specific product or use case for this mouthpiece.*
Figure F - advairhfa spl graphic 12

The given text states a simple instruction to breathe out as much air as possible.*
Advair HFA 115mcg 120 dose carton - advairhfa spl graphic 17

This is a description of a medication called ADVAIR HFA, which is intended for oral inhalation and contains fluticasone propionate and salmeterol xingfoate in a microcrystalline suspension. The medication is delivered in doses of 45 mcg of fluticasone propionate and 3045 mcg of sameterol xinafoate, which is equivalent to 21 meg ofsalmeterol base, using an ADVAIR HFA actuator. The text provides NDC number and advises to refer to prescribing information for dosage details.*
Advair HFA 230mcg 120 dose carton - advairhfa spl graphic 18

This is a description of ADVAIR HFA, an inhalation aerosol used for oral inhalation. It contains a microcrystalline suspension of fluticasone propionate and salmeterol xinafoate in propellant HFA-134a. Each dose delivers 115 mcg of fluticasone propionate and 304.5 mcg of salmeterol xinafoate (equivalent to 21 mcg of salmeterol base) from the mouthpiece. The prescribing information should be consulted for dosage information.*
advairhfa spl graphic 19

This is the description: ADVAIR HFA is an inhalation aerosol containing a microcrystalline suspension of Fluticasone propionate and Salmeterol. It is used for oral inhalation with the ADVAIR HFA actuator only. Each canister contains 230 mcg of Fluticasone propionate and 3045 mcg of Salmeterol equivalent to 21 mcg of Salmeterol base from the mouthpiece. Please refer to the prescribing information for dosage instructions.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.