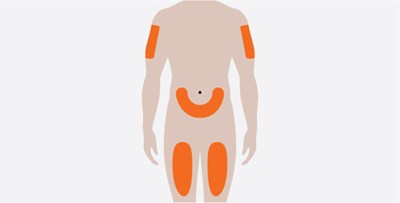
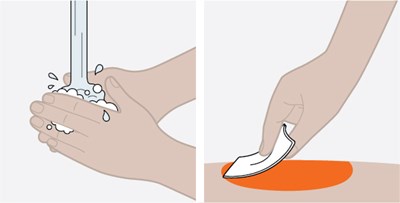
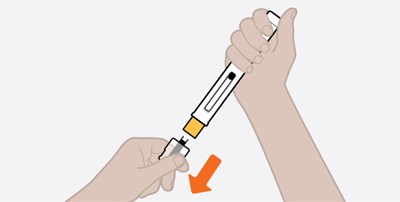
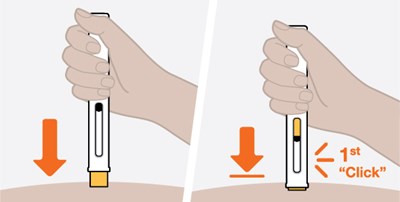
Product Images Nucala
View Photos of Packaging, Labels & Appearance
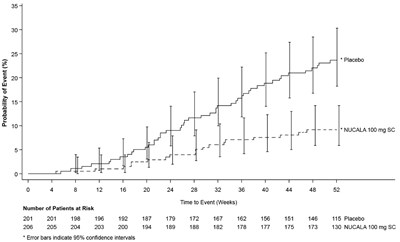
- Figure 1. Kaplan-Meier Cumulative Incidence Curve for Time to First Exacerbation (Trial 2) - nucala spl graphic 01
- Figure 2. Kaplan-Meier Plot of Time to First Relapse - nucala spl graphic 02
- Nucala 100 mg per vial carton - nucala spl graphic 03
- nucala spl graphic 04
- nucala spl graphic 05
- nucala spl graphic 06
- nucala spl graphic 07
- nucala spl graphic 08
- nucala spl graphic 09
- nucala spl graphic 10
- nucala spl graphic 11
- nucala spl graphic 12
- nucala spl graphic 13
- nucala spl graphic 14
- nucala spl graphic 15
- nucala spl graphic 16
- nucala spl graphic 17
- nucala spl graphic 18
- nucala spl graphic 19
- nucala spl graphic 20
- nucala spl graphic 21
- nucala spl graphic 22
- nucala spl graphic 23
- nucala spl graphic 24
- nucala spl graphic 25
- nucala spl graphic 26
- nucala spl graphic 27
- nucala spl graphic 28
- nucala spl graphic 29
- nucala spl graphic 30
- nucala spl graphic 31
- nucala spl graphic 32
- nucala spl graphic 33
- nucala spl graphic 34
Product Label Images
The following 34 images provide visual information about the product associated with Nucala NDC 0173-0881 by Glaxosmithkline Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. Kaplan-Meier Cumulative Incidence Curve for Time to First Exacerbation (Trial 2) - nucala spl graphic 01

This appears to be a graph displaying the probability of an event for different treatments over a period of weeks. The treatments include a placebo, Mepolizumab 75 mg IV, and NUCALA 100 mg SC. The graph also indicates the number of patients at risk and the number of events occurring over the weeks. There are error bars indicating 95% confidence intervals.*
Figure 2. Kaplan-Meier Plot of Time to First Relapse - nucala spl graphic 02
This appears to be a graph showing the probability of an event over time for two treatments. The treatments are "Placebo" and "NUCALA 100 mg SC". The x-axis represents time in weeks, while the y-axis represents the probability of the event occurring in percentage. The graph also shows error bars indicating 95% confidence intervals. However, no information is available on what the specific event being measured is.*
Nucala 100 mg per vial carton - nucala spl graphic 03

This is a graph showing the probability of an event in percentage for two groups, placebo and NUCALA 300 mg SC. The x-axis shows the time to event in weeks, and the y-axis shows the probability of the event in percentage. The graph also includes the number of patients at risk for each group. Error bars indicating 95% confidence intervals are also shown.*
nucala spl graphic 04

This is a graph of the probability of an event (measured in percent) over time (measured in weeks) for patients given a placebo versus those given NUCALA 300 mg SC. The graph shows the number of patients at risk over time for each treatment group, and error bars indicate 95% confidence intervals.*
nucala spl graphic 05
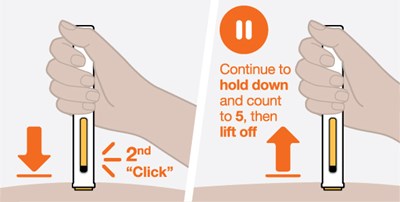
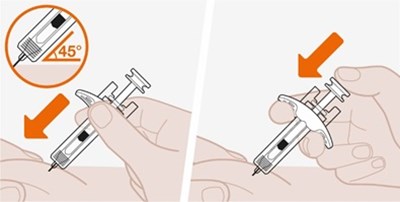
The given text is a set of instructions for a medical injection device that includes information about how to use the device before and after use. It mentions the label, stopper, inspection window, needle guard, needle cap, and yellow indicator. The yellow indicator becomes visible once the injection is completed.*
nucala spl graphic 06
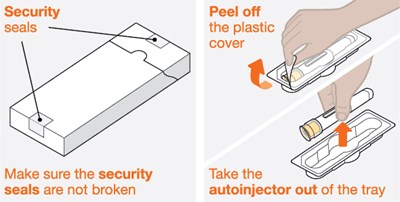
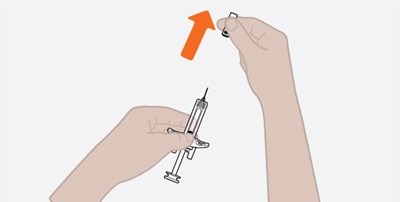
This is a description regarding the security of an autoinjector. The text suggests that there are security peel-off seals that cover the plastic cover of the autoinjector. It further emphasizes the importance of those seals not being broken. Finally, it recommends taking the autoinjector out of its tray.*
nucala spl graphic 07

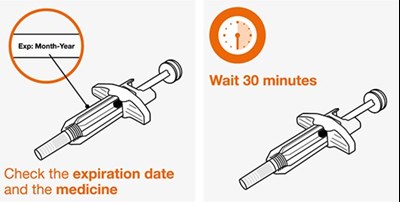
This text is a simple instruction advising to check the expiration date and whether it is a medicine.*
nucala spl graphic 13

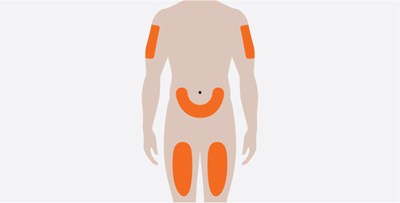
This text indicates the presence of a container for the disposal of sharps such as needles, lancets, or blades. No further information is available.*
nucala spl graphic 14
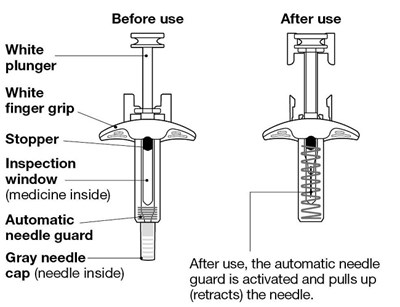
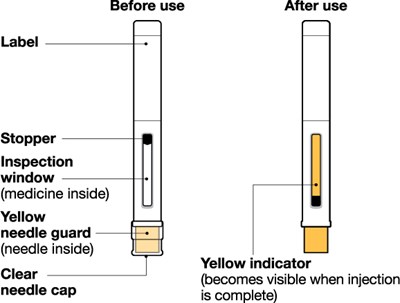
The text describes a medical device consisting of a white plunger with a white finger grip and a gray needle. It also has a stopper and an inspection window to check the medicine inside. After use, an automatic needle guard is activated, pulling the needle inside a cap.*
nucala spl graphic 15

This text appears to be a set of instructions related to maintaining the security of a plastic cover for a syringe. It advises the reader to ensure that the "security seals" on the cover are not broken, and to remove the syringe from its tray.*
nucala spl graphic 21

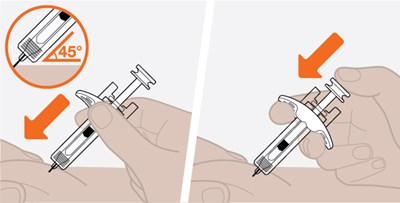
The text seems to be a simple instruction. It says "Make sure white plunger is fully down". It appears to be a direction for a user to ensure that a particular plunger is entirely pressed downwards.*
nucala spl graphic 23

This is a description of a medical syringe. It has a white plunger and a white finger grip, and there is an inspection window to see the medicine inside. The syringe also has an automatic needle guard and a gray needle cap, which retracts the needle after use.*
nucala spl graphic 24
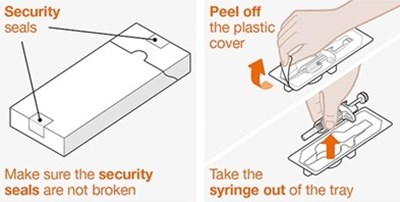
This text provides instructions on how to secure a plastic cover using a security peel-off seal. It also advises taking the syringe out of the tray while ensuring that the security seals are not broken.*
nucala spl graphic 25

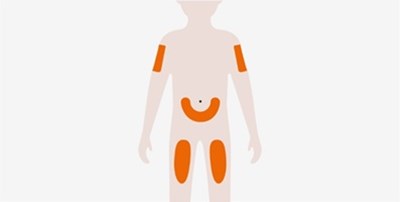
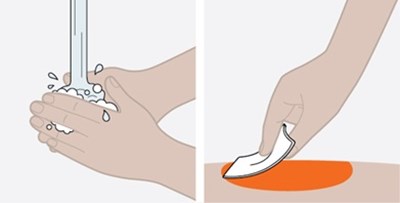
This text provides basic instructions for taking medication. It advises the reader to check the expiration date of the medicine before taking it, as well as to wait 30 minutes after taking it.*
nucala spl graphic 30
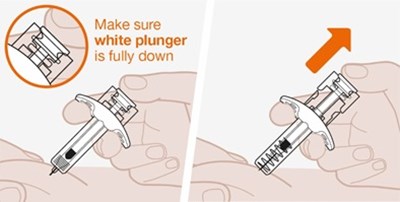
A manual instruction or warning to ensure that a white plunger is pushed completely down. There is not enough context to determine the specific purpose or product related to the instruction.*
nucala spl graphic 31

This text simply states the name of a product, which is a "Sharps Disposal Container". It is likely used for safe and proper disposal of sharp medical tools or needles.*
nucala spl graphic 32

Nucala is a prescription drug in the form of an injection used after reconstitution. The reconstituted solution has a concentration of 100 mg/mL. It is supplied in a single-dose vial with no preservatives and should be discarded if any portion is unused. Each vial contains 100 mg of mepolizumab, along with polysorbate 80, sodium phosphate dibasic heptahydrate, and Sucrose. Nucala is strictly for subcutaneous use only with no potency standard in the United States.*
nucala spl graphic 34

Nucala (mepolizumab) Injection 40mg/0.4mL Prefilled Syringe is a single-dose, subcutaneous medication used for prescription only. It comes with a prefilled syringe, along with instructions for use, prescribing, and patient information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.