Product Images Heparin Sodium In Sodium Chloride
View Photos of Packaging, Labels & Appearance
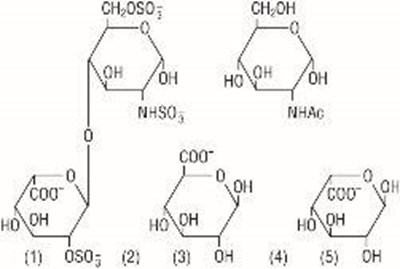
- heparin sodium chemical formula - heparin sodium in sodium chloride injection in exc 1
- =4 - heparin sodium in sodium chloride injection in exc 2
- Recycle 7 - heparin sodium in sodium chloride injection in exc 3
- Heparin Sodium in 0.45% Sodium Chloride 25,000 units per 500mL - heparin sodium in sodium chloride injection in exc 4
- =4 - heparin sodium in sodium chloride injection in exc 5
- Recycle 7 - heparin sodium in sodium chloride injection in exc 6
- Heparin Sodium in 0.45% Sodium Chloride 25,000 units per 250mL - heparin sodium in sodium chloride injection in exc 7
Product Label Images
The following 7 images provide visual information about the product associated with Heparin Sodium In Sodium Chloride NDC 0264-3462 by B. Braun Medical Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Heparin Sodium in 0.45% Sodium Chloride 25,000 units per 500mL - heparin sodium in sodium chloride injection in exc 4

Description: This text provides detailed information about the injection solution containing Heparin Sodium and 0.45% Sodium Chloride. Each 100mL of the solution consists of 5,000 USP Heparin units, Sodium Chloride, Dibasic Sodium Phosphates, and Citic Acid Anhydrous among other components. It gives electrolyte values and warns against admixing with other drugs. The solution is for intravenous use only and comes in single-dose containers. It should be stored at controlled temperatures and protected from freezing. The text also includes prescribing information and details about the product's composition and manufacturing.*
Heparin Sodium in 0.45% Sodium Chloride 25,000 units per 250mL - heparin sodium in sodium chloride injection in exc 7

This text appears to be a label for a medical product, specifically Heparin USP units mixed with a 0.45% Sodium Chloride solution for intravenous use. It provides information on the contents per 100 mL, including Heparin Sodium USP, Sodium Chloride USP, Dibasic Sodium Phosphate, Citric Acid Anhydrous, and Water for Injection. The label also includes electrolyte amounts in milliequivalents per 100 mL. The text warns not to mix with other drugs, specifies storage conditions, and instructs users to refer to the Prescribing Information for dosage. Additionally, it mentions that it is a sterile, single-dose container that should be discarded if unused, and not to remove the overwrap until ready for use.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.