Product Images Heparin Sodium
View Photos of Packaging, Labels & Appearance
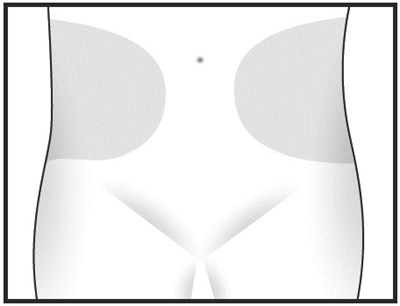
- Chemical structure - heparin sodium injection syringe 1
- step-8 - heparin sodium injection syringe 10
- blister label - heparin sodium injection syringe 11
- pack label - heparin sodium injection syringe 12
- syringe label - heparin sodium injection syringe 13
- syringe - heparin sodium injection syringe 2
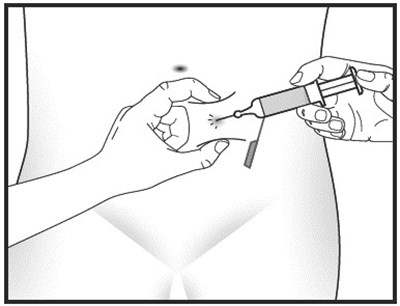
- step-1 - heparin sodium injection syringe 3
- step-2 - heparin sodium injection syringe 4
- step-3 - heparin sodium injection syringe 5
- step-4 - heparin sodium injection syringe 6
- step-5 - heparin sodium injection syringe 7
- step-6 - heparin sodium injection syringe 8
- step-7 - heparin sodium injection syringe 9
Product Label Images
The following 13 images provide visual information about the product associated with Heparin Sodium NDC 0264-5705 by B. Braun Medical Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
blister label - heparin sodium injection syringe 11

This is a description of a prescription drug called "Sodium Injection, PreservativeRe_ 0 e 1" made by B/BRAUN. It contains 5,000 USP units/0.5 ML for intravenous use. The drug is packaged in a single dose prefilled syringe and it should not be used for lock flush. The medication should be injected into the intestinal mucosa. The expiration date is not provided.*
pack label - heparin sodium injection syringe 12

This is a product label for Heparin sodium injection. The injection contains 5,000 USP units per 0.5 mL and is intended for subcutaneous or intravenous use. The label includes instructions for use and precautions for handling the medication. The medicine should not be used for lock flush. The product is sold in pre-filled syringes.*
syringe - heparin sodium injection syringe 2

This appears to be a diagram or image labeling different parts of a syringe. The labeled parts include the needle shield, syringe body, needle itself, plunger, protective needle cap, plunger rod, and back stop. No further context or information is available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.