Product Images Byetta
View Photos of Packaging, Labels & Appearance
- 5mcg cannot be pushed in
- 5mcg window
- Arrow for dose knob
- Arrow in dose window cannot be pushed in
- Arrow knob wont turn
- Injection arrow
- Injection complete arrow
- Note 5mcg
- Pull dos 5mcg
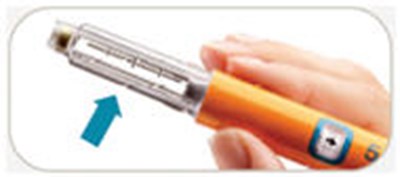
- Pull out arrow
- Reset 5mcg arrow
- Step 3 arrow 5mcg
- Step 3 arrow dose 5mcg
- Step E arrow 5mcg
- Turn dos 5mcg
- Turn dose 5mcg 01
- Turn knob away arrow
- Figure 1 - byetta 01
- Figure 2 - byetta 02
- Figure 3 - byetta 03
- Byetta parts - byetta 05
- Byetta Pen Parts and Dose Window Symbols - byetta 06
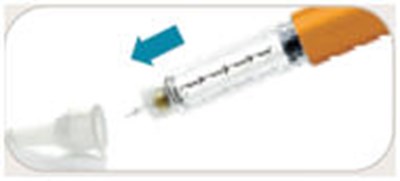
- Remove paper from needle - byetta 09
- Pull off outer needle - byetta 10
- Pull off inner needle shield - byetta 11
- Arrow 4 - byetta 17
- Point needle away from you - byetta 18
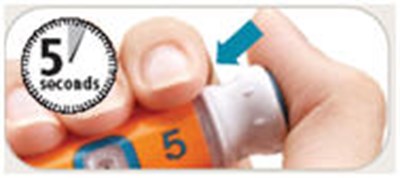
- Push and Hold - byetta 19
- Pen preparation - byetta 20
- Check pen label - byetta 22
- Check cartridge - byetta 23
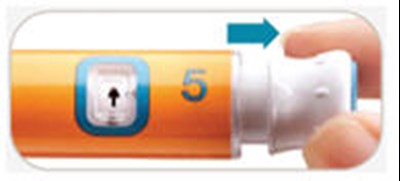
- Check window dose - byetta 27
- Pull dose knob - byetta 28
- Grip pen firmly - byetta 30
- Push and hold - byetta 31
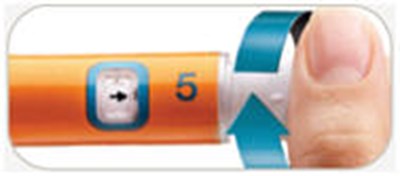
- Injection is complete - byetta 32
- Turn dose window - byetta 33
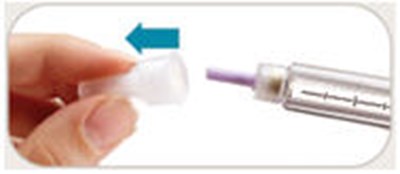
- Carefully put the outer needle - byetta 34
- Unscrew the needle - byetta 35
- Throw away needles - byetta 36
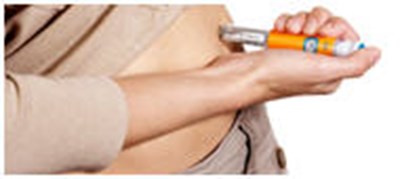
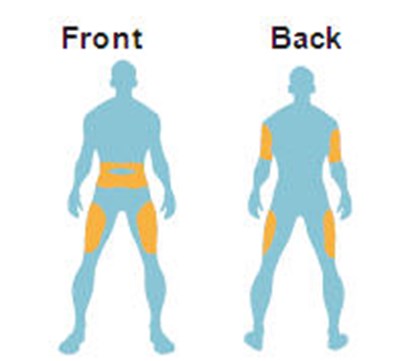
- Where to inject Byetta - byetta 37
- 2.4 mL Cartridge Carton - byetta 70
- 1.2 mL Cartridge Carton - image 01
- image 02
- image 03
- image 04
- image 05
- image 06
- image 07
- image 08
- image 09
- image 10
- image 11
- image 12
- image 13
- image 14
- image 15
- image 16
- image 17
- image 18
- image 19
- image 20
- image 21
- image 22
- image 23
- image 24
- image 25
- image 26
- image 27
- image 28
- image 29
- image 30
- image 31
- image 32
- image 33
- image 34
- image 35
- image 36
- image 37
- image 38
- image 39
- image 40
- image 41
- image 42
- image 43
- image 44
- image 45
- image 46
- image 47
- image 48
- image 49
- image 50
- image 51
- image 52
- image 53
- image 54
- image 55
- image 56
- image 57
- image 58
Product Label Images
The following 100 images provide visual information about the product associated with Byetta NDC 0310-6512 by Astrazeneca Pharmaceuticals Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - byetta 01
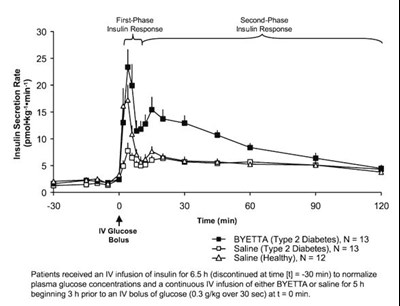
This is a chart showing the insulin response in patients with Type 2 Diabetes who received an IV infusion of insulin, followed by either BYETTA or saline for 6.5 hours prior to an IV bolus of glucose. The chart shows glucose levels over time for the three groups of patients: BYETTA-treated Type 2 Diabetics, saline-treated Type 2 Diabetics, and saline-treated healthy patients. The study aimed to evaluate the effects of the different treatments on insulin response.*
Figure 2 - byetta 02
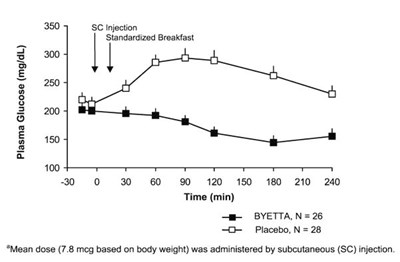
This text describes a study or experiment involving a injection called "350 SC Injection". A standardized breakfast was likely given to the participants. The rest of the text consists of numerical data and a graph showing "BYETTAN" and "Placebo" on a time scale in minutes. Mean dose administered was 7.8 mog based on body weight via subcutaneous injection.*
Figure 3 - byetta 03
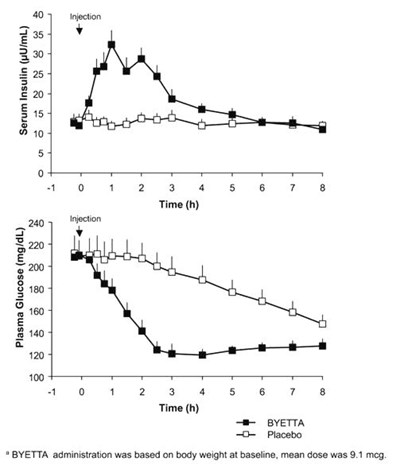
This is a medical report that shows the measurements of Plasma Glucose and Serum Insulin levels in mg/dL and 4U/mL respectively over time (in hours). The report also mentions the administration of BYETTA and Placebo based on the participant's body weight.*
Byetta Pen Parts and Dose Window Symbols - byetta 06

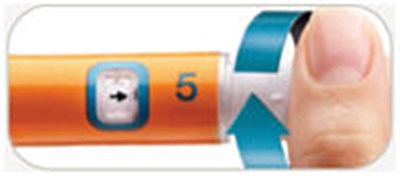
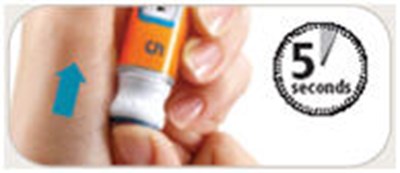
This appears to be a set of instructions related to a medical injection device. It specifies different states the device can be in and what actions can be taken in each state. It mentions a dose knob and injection of 5 mg, but does not provide any specific details on what medication is being administered. The inclusion of "Pen Needles Not Included" suggests that this may be a disposable device that requires separate needles to be attached.*
2.4 mL Cartridge Carton - byetta 70

This is a medication for subcutaneous use only, which contains 60 doses of 10mg each. It should be refrigerated and not frozen. The medication cannot be transferred to a syringe, and pen needles are not included. The user should consult their healthcare provider regarding the appropriate needle size and gauge to use, and it is recommended to use disposable pen needles with 28, 30, or 31 gauge. The medication is called Revazeoccs®.*
1.2 mL Cartridge Carton - image 01

This is a medication label for DG aat0-6812.01 which contains medication to be dispensed to patients. The medication contains 250 mg/mL and comes in a 12 mL package. Each prefilled pen contains 60 doses of the medication and is meant for subcutaneous use only. It should be refrigerated and not frozen. The medication should not be transferred to a syringe. The medication label mentions that the pen needles are not included and advises patients to consult their healthcare provider to determine the best length and gauge of a disposable pen needle. The recommended pen needle gauges are 23 (i), 30, or 31 (thinner).*
image 13

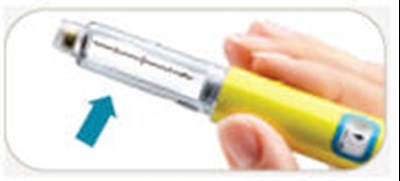
This is a list of parts for a BYETTA pen used for injecting medication. It includes a blue pen cap, cartridge, BYETTA label, dose window, dose knob, and injection button.*
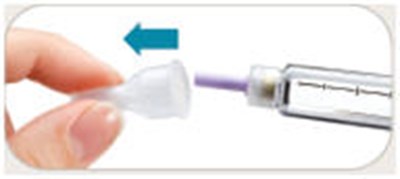
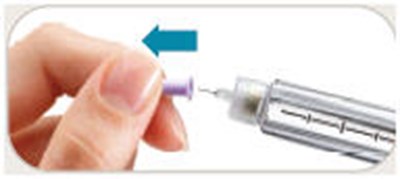
image 14

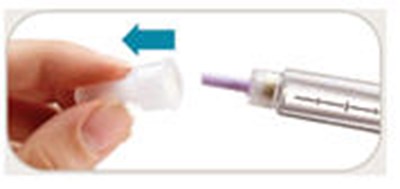
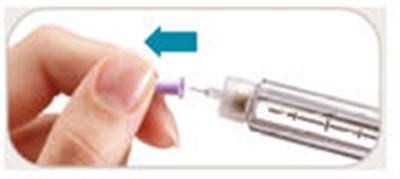
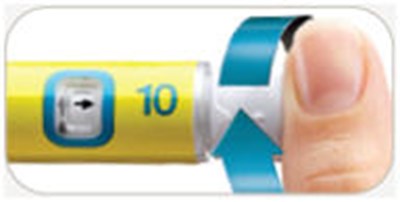
This is a description of the parts of a pen needle, excluding the pen needles themselves. The parts include an outer needle, inner needle, needle shield, and paper tab. The text also mentions symbols on a dose window that indicate when the dose knob is ready to be pulled out, ready to be turned to the dose position, and when it is ready to inject a 10-meg dose. Additionally, there is a symbol that indicates when the dose knob has been pushed in and is ready to reset.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.