Product Images Budesonide And Formoterol Fumarate Dihydrate
View Photos of Packaging, Labels & Appearance
- Counter Image 15 - 308708 rapihaler counter 15 (black;130;485)
- Counter Image 80 - 308710 rapihaler counter 80 (black;130;485)
- Figure 1 - figure1bodytext
- Figure 2 - figure2bodytext
- Figure 3 - figure3bodytext
- Figure 4 - figure4bodytext
- Figure 5 - figure5bodytext
- Figure 6 - figure6bodytext
- Figure 7 - figure7bodytext
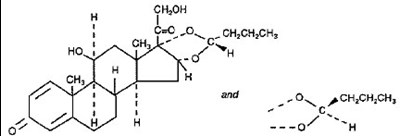
- budesonide structural formula - image 01
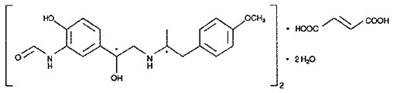
- structural fumarate - image 02
- Figure 1 - image 03
- Figure 2 gray arrows - image 04
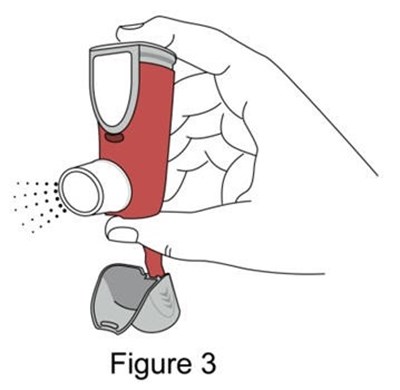
- Figure 3 - image 05
- Figure 4 - image 06
- Figure 5 - image 07
- Symbicort AG 80/4.5 mcg 120 inhalations carton - image 10
- Symbicort AG 160/4.5 mcg 120 inhalations carton - image 11
Product Label Images
The following 18 images provide visual information about the product associated with Budesonide And Formoterol Fumarate Dihydrate NDC 0310-7372 by Astrazeneca Pharmaceuticals Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - figure1bodytext

This appears to be a medical document containing a table of numbers, with titles including "Treatment" and various medication names and doses. One medication mentioned is Budesonide and Formoterol Fumarate Dihydrate, with recommended dosages listed.*
Figure 4 - figure4bodytext

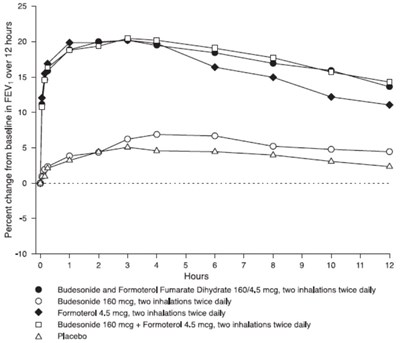
This is a graph that shows the percent change from baseline in FEV (Forced Expiratory Volume) over 12 hours. It includes data for 12 hours on day 1 of a study using Budesonide and Formoterol Fumarate Dihydrate inhalations for treatment. The data points are shown at multiple time intervals, marked on the x-axis in hours. The graph also includes a placebo measurement, marked as 'A Placebo' in the Predose FEV column.*
Figure 6 - figure6bodytext

This text provides data on the percent change from baseline in pre-dose FEV (forced expiratory volume) for various treatments involving Budesonide and Formoterol Fumarate Dihydrate MDI, with dosage and inhalation frequency specified. The data includes numerical values for each treatment and a placebo control.*
Figure 7 - figure7bodytext

This is a table showing the percent change from baseline in 1-hour post-dose FEV with different inhalations of Budesonide and Formoterol Fumarate Dihydrate, Budesonide, Formoterol, Budesonide + Formoterol, and Placebo over a 6-months period. The text also includes the dosages and frequencies for each inhalation.*
Figure 1 - image 03

This text is a diagram or image labeling related to a counter with a canister inside containing water, a mouthpiece with a strap, and a mouthpiece cover.*
Symbicort AG 80/4.5 mcg 120 inhalations carton - image 10

This is a description of a medication called Budesonide and Formoterol Fumarate Dihydrate Inhalation Aerosol. The medication comes in a canister with 120 inhalations and has the NDC code 0310-7372:20.*
Symbicort AG 160/4.5 mcg 120 inhalations carton - image 11

This is a description of a medication with the name "Budesonide and Formoterol Fumarate Dihydrate Inhalation Aerosol 160/4.5". It consists of Budesonide and Formoterol Fumarate Dihydrate in the ratio of 160 mcg and 4.5 mcg respectively. One container has 120 inhalations and is meant for oral inhalation only. It is a prescription medicine produced in France by AstraZeneca. Its NDC number is 0310-7376-20.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
.jpg)
.jpg)