Product Images Airsupra
View Photos of Packaging, Labels & Appearance
- Albuterol structural formula - image 01
- Budesonide structural formula - image 02
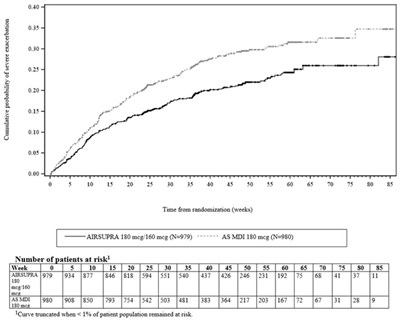
- Figure 1 - image 03
- Parts of your inhaler - image 04
- Reading the puff indicator - image 05
- Write the date - image 06
- 1-Prime your inhaler before first use - image 07
- 2-Prime your inhaler before fist use - image 08
- When and how to Re-prime your inhaler - image 09
- Check - image 10
- 1-Shake and Inhale - image 11
- 2-Shake and Inhale - image 12
- 3-Shake and Inhale - image 13
- 4-Shake and Inhale - image 14
- 5-Shake and Inhale - image 15
- Repeat - image 16
- Place cover back on - image 17
- Rinse - image 18
- Remove - image 19
- 1-Rinse through both ends - image 20
- 2-Rinse through both ends - image 21
- Air-dry - image 22
- Re-assemble and Re-prime - image 23
- Parts of your inhaler - image 24
- Reading the puff indicator - image 25
- Write the date - image 26
- 1-Prime your inhaler before first use - image 27
- 2-Prime your inhaler before fist use - image 28
- When and how to Re-prime your inhaler - image 29
- Check - image 30
- 1-Shake and Inhale - image 31
- 2-Shake and Inhale - image 32
- 3-Shake and Inhale - image 33
- 4-Shake and Inhale - image 34
- 5-Shake and Inhale - image 35
- Repeat - image 36
- Place cover back on - image 37
- Rinse - image 38
- Remove - image 39
- 1-Rinse through both ends - image 40
- 2-Rinse through both ends - image 41
- Air-dry - image 42
- Re-assemble and Re-prime - image 43
- Principal Display Panel - Carton Label - image 44
Product Label Images
The following 44 images provide visual information about the product associated with Airsupra NDC 0310-9080 by Astrazeneca Pharmaceuticals Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Parts of your inhaler - image 04

The text describes the components of an inhaler. It includes a puff indicator, a pressurized canister that holds the medicine, an actuator that contains the canister, a mouthpiece that sprays the medicine, and a mouthpiece cover to protect it when not in use. The label on the actuator should have a "Use By Date" written on it.*
Reading the puff indicator - image 05

This text appears to be information about using an inhaler. The "pointer" is used to indicate the number of puffs remaining in the inhaler. When the pointer is in the yellow zone, it is a signal to order a new inhaler. If the pointer reaches 0 in the red zone, it is recommended to dispose of the inhaler.*
Parts of your inhaler - image 24

This text appears to be a description of an inhaler device and its components. The inhaler consists of a pressurized canister filled with medicine, which is held inside an actuator. The actuator has a mouthpiece, which sprays the medicine when used. There is also a mouthpiece cover to protect the mouthpiece when the inhaler is not in use. The text also mentions a puff indicator, which is attached to the top of the pressurized canister. Finally, it suggests writing the "Use By Date" on the label of the actuator.*
Reading the puff indicator - image 25

This text appears to be instructions or information related to an inhaler device. It mentions a "pointer" that indicates the number of puffs remaining. There are two zones mentioned, a yellow zone and a red zone. When the pointer is in the yellow zone, it suggests ordering a new inhaler. In the red zone, it advises throwing away the inhaler when the pointer reaches 0.*
Principal Display Panel - Carton Label - image 44

Discard the inhaler when either 12 months have passed since removing it from the foil pouch, or after the expiration date on the carton and canister, or when the dose indicator shows 0, whichever comes first. For more information, visit the website www.AIRSUPRA.com or use your mobile phone to scan the QR code provided. The inhaler is called AIRSUPRA™ and it contains albuterol and budesonide. It is an Inhalation Aerosol meant for oral inhalation only. Each inhalation contains 90 mcg of albuterol and 80 mcg of budesonide. The inhaler should be shaken well before using and contains 120 inhalations. This product is manufactured by AstraZeneca.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.