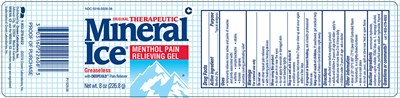
FDA Label for Mineral Ice Pain Relieving
View Indications, Usage & Precautions
- ACTIVE INGREDIENT
- PURPOSE
- USES
- WARNINGS
- DO NOT USE
- WHEN USING THIS PRODUCT
- STOP USE AND ASK A DOCTOR IF
- IF PREGNANT OR BREAST-FEEDING
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS OR COMMENTS?
- ADDITIONAL INFORMATION LISTED ON OTHER PANELS
- PRINCIPAL DISPLAY
Mineral Ice Pain Relieving Product Label
The following document was submitted to the FDA by the labeler of this product Crown Laboratories. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient
Menthol 2%
Purpose
Topical analgesic
Uses
● temporarily relieves minor aches and pains of muscles and joints associated with:
● arthritis ● simple backache ● strains
- ● bruises ● sprains
● provides cooling penetrating relief
Warnings
For external use only
Do Not Use
- with other topical pain relievers
- with heating pads or heating devices
When Using This Product
- do not use in or near the eyes
- do not apply to wounds or damaged skin
- do not bandage tightly
Stop Use And Ask A Doctor If
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
If Pregnant Or Breast-Feeding
ask a health professional before use
Keep Out Of Reach Of Children
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- clean affected area before applying product
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
Other Information
- store at 20 o - 25 oC (68 o - 77 oF) [see USP Controlled Room Temperature], in a tightly closed container
- store in a cool place
- do not use, pour, spill or store near heat or open flame
Inactive Ingredients
ammonium hydroxide, carbomer, cupric sulfate, FD&C blue no. 1, isopropyl alcohol, magnesium sulfate, purified water, sodium hydroxide, thymol
Questions Or Comments?
call 1-833-279-6522
Additional Information Listed On Other Panels
Distributed by: Crown Laboratories, Inc.
Johnson City, TN 37604 ©2018 Made in Canada.
ORIGINAL THERAPEUTIC
Mineral Ice ®
Menthol Pain Relieving Gel
Greaseless with DEEPCOLD ® Pain Reliever
Mineral Ice and the Mineral Ice Mountain range are registered trademarks of Crown Laboratories, Inc.
Principal Display
0316-0226-08
Mineral Ice®
Menthol Pain Relieving Gel
Greaseless with DEEPCOLD® Pain Reliever
Net wt. 8 oz (226.8 g)
Distributed by: Crown Laboratories, Inc.
Johnson City, TN 37604
p11529.00
* Please review the disclaimer below.