Product Images Doxil
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Doxil NDC 0338-0067 by Baxter Healthcare Corporation, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - image 05

This appears to be a statistical report and includes some kind of medical intervention. There are mentions of a treatment called Bortezomib and a p-value of less than
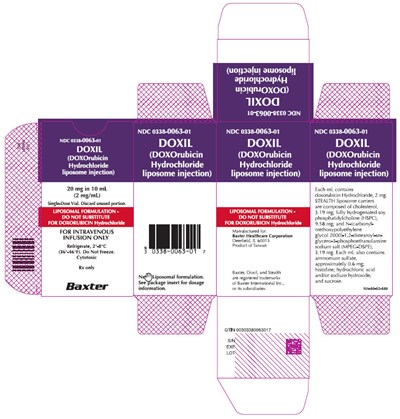
Representative 20 mg Container Label - image 06

This is a description of a medication with the NDC code 0338-0063-01. It is a liposome injection for intravenous infusion, with 20 mg in 10 mL (2 mg/mL) dosage. It is a single-dose vial and the unused portion should be discarded. This medication is a LIPOSOMAL FORMULATION, and it is important not to substitute it for doxorubicin Hydrochloride. It is to be used under prescription only and is cytotoxic. It should be refrigerated between 36°-46°F, but not frozen. Dosage information can be found in the package insert. It is manufactured by Baxter Healthcare Corporation located in Deerfield, IL 60015.*
Representative Container Label 50 mg - image 09
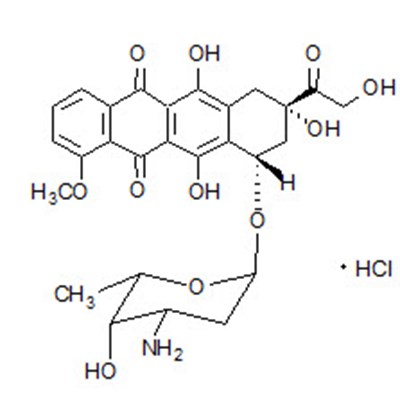
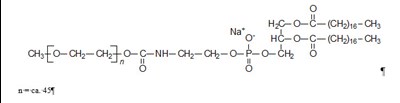
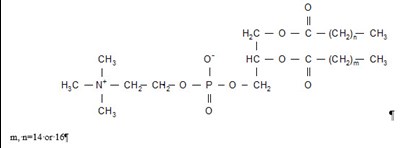
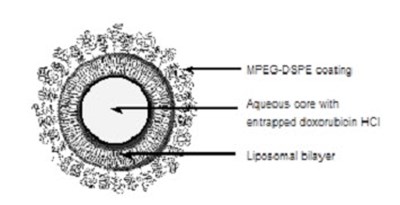
DOXIL is a liposomal formulation of DOXOrubicin Hydrochloride injection used for intravenous infusion only. Each 10mL vial contains 20mg of DOXOrubicin Hydrochloride. It should not be substituted for other DOXOrubicin Hydrochloride products. The drug should be refrigerated at 2.5°C and should not be frozen. The liposomes used in DOXIL carries doxorubicin hydrochloride and are composed of cholesterol, ycropensted s0y phosphathholin 15, Necrbonyle methanypolyetyien and others. The concentration of doxorubicin hydrochloride in the injection is approximately 2mg/mL. The drug is only available in a 10mL vial and should be administered by a licensed healthcare provider.*
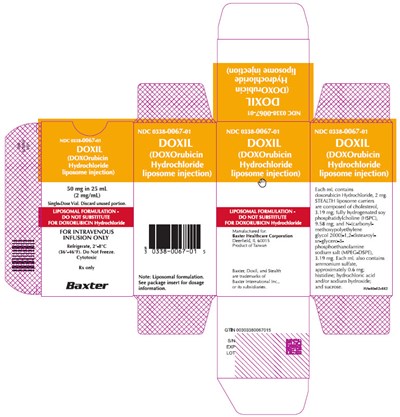
Representative Carton Label 0338-0067-01 - image 10

This is a medication with the National Drug Code (NDC) 0338-0067-01. It needs to be refrigerated between 2°-8°C (36°46°P) and should be discarded if not used completely. It contains a liposomal formulation of 50mg in 25 mL of Doxorubicin Hydrochloride, which is used for intravenous infusion only as a cytotoxic. The text includes a warning not to substitute it for regular Doxorubicin. There is also a reference to a package insert for dosage information.*
Representative Carton Label 50 mg Taiwan - image 11

This text appears to be a combination of a medical prescription and a packaging label for a drug called DOXIL, which is a liposome injection. The label indicates that the drug should be diluted with 5% Dextrose Inecton, USP and discarded if unused. It also warns against substituting DOXIL with DOXORUBICIN Hydrochloride and recommends storage between 2-8°C and not to freeze. However, there is no information about the correct use, indications, or side effects of this medication.*
image 12

DOXIL is a liposome injection containing doxorubicin Hycrochloride, used for the treatment of ovarian and breast cancer. The liposome carriers provide increased effectiveness and reduce side effects. It is essential not to use DOXIL as a substitute for DOXORUBICIN Hydrochloride. The drug is manufactured and marketed by Baxter Healthcare in Deerfield, IL. The injection has a unique composition and should only be used as directed by a physician.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.