Product Images Clindamycin Phosphate
View Photos of Packaging, Labels & Appearance
- Representative Container NDC 0338-3814-50 panel 2 of 2 - Image 13
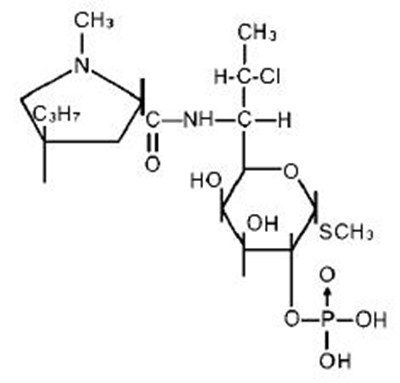
- Clindamycin in 5% Dextrose Structural Formula - image 01
- Representative Container Label NDC 0338-3410-50 panel 1 of 2 - image 02
- Representative Container Label NDC 0338-3410-50 panel 2 of 2 - image 03
- Representative Carton NDC 0338-3410-24 panel 1 of 2 - image 04
- Representative Carton NDC 0338-3410-24 panel 2 of 2 - image 05
- Representative Serialized Label 0338-3410-24 - image 06
- Representative Container NDC 0338-3612-50 panel 1 of 2 - image 07
- Representative Container NDC 0338-3612-50 panel 2 of 2 - image 08
- Representative Carton NDC 0338-3612-24 panel 1 of 2 - image 09
- Representative Carton NDC 0338-3612-24 panel 2 of 2 - image 10
- Representative Serialized Label 0338-3612-24 - image 11
- Representative Container NDC 0338-3814-50 panel 1 of 2 - image 12
- Representative Carton NDC 0338-3814-24 panel 1 of 2 - image 14
- Representative Carton NDC 0338-3814-24 panel 2 of 2 - image 15
- Representative Serialized Label 0338-3814-24 - image 16
Product Label Images
The following 16 images provide visual information about the product associated with Clindamycin Phosphate NDC 0338-3612 by Baxter Healthcare Company, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Representative Container Label NDC 0338-3410-50 panel 1 of 2 - image 02

Clindamycin Injection USP is a prescription medication that is only available for intravenous use. It comes in a single-dose container with 5% Dextrose and the quantity of the medicine is 300 mg per 50 mL of injection. The medication is indicated for intravenous use only and must not be given through other series connections. It is advised to check for minute leaks and the clarity of the solution before using it. If the solution is not clear, it cannot be used. It is also advised to discard any unused portion of the injection. The dosage should be as directed by a doctor, and the prescribing information should be consulted before use. The medication must not be administered in a rapid injection or by Intravenous push. The injection contains Clindamycin phosphite, USP equivalent to 300 mg Clindamycin, 2.5 g Dextrose hydrous, USP, 2 mg Edetate disodium, IP, USP, and water for injection, USP. The pH may have been adjusted with sodium hydroxide and/or hydrochloric acid. The injection should be stored at 20°C to 25°C (68° to 77°F), and temperatures above 30°C should be avoided.*
Representative Carton NDC 0338-3410-24 panel 1 of 2 - image 04

This is a description of a product called "Galaxy Container" made by Baxter Healthcare Corporation. The container is made of PL 2501 plastic material and is produced in the USA. The text also mentions a trademark and the manufacturer's address. Additionally, there is information about a medicine called "Clindamycin Injection USP" in 5% dextrose. The medicine comes in 50mL single dose containers containing 300mg per 50mL. The text provides information on how to store the medicine and advises avoiding high temperatures.*
Representative Carton NDC 0338-3410-24 panel 2 of 2 - image 05

This is a prescription-only medication for intravenous use that contains 300mg of Clindamycin phosphate and other ingredients such as dextrose, sodium hydride, and water for injection. It is nonpyrogenic and should not be injected rapidly or used in series connections. Dosage and cautionary information are available in the prescribing information.*
Representative Carton NDC 0338-3612-24 panel 1 of 2 - image 09

This is a description of medical containers and their contents. The first section refers to a plastic container made in the USA and trademarks of a healthcare company. The second section talks about Clindamycin Injection USP in 5% Dextrose, including dosage information and storage instructions.*
Representative Carton NDC 0338-3612-24 panel 2 of 2 - image 10

This text appears to be a description of a medication that is intended for intravenous use only. It contains specifications about the contents of the medication and information on dosage and cautions. The medication is nonpyrogenic, and caution is advised to check for minute leaks and solution clarity. However, the actual name of the medication is not mentioned.*
Representative Carton NDC 0338-3814-24 panel 1 of 2 - image 14

This is a description of a medical product called GALAXY Container PL2501 Plastic Clindamycin Injection USP in 5% Dextrose. The product is made in the USA and contains 12-50mL single dose containers. The dosage is 900mg per 50mL of solution (18mg/mL). The product is a registered trademark of Baxter International and Baxter Healthcare Corporation. The recommended storage temperature is controlled room temperature and should not exceed a certain temperature limit.*
Representative Carton NDC 0338-3814-24 panel 2 of 2 - image 15

This is a description of a medication that is available only by prescription. The medication is for intravenous use, is sterile and nonpyrogenic, and should not be administered via rapid injection or intravenous push. Each 50 ml of the medication contains a specific amount of Gindamycin phosphate and dextrose, along with other ingredients. The pH of the medication may have been adjusted using sodium hydroxide or hydrochloric acid. The dosage information is available in the prescribing information, and caution should be exercised to avoid adding supplementary medication or using it in series connections. The solution must also be carefully checked for minute leaks.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.