Product Images Tisseel Fibrin Sealant
View Photos of Packaging, Labels & Appearance
- Figure 1: DUO SET - image 01
- Tisseel Frozen 2mL Representative Pouch Label - NDC 0338-8402-01 - image 02
- Tisseel Frozen 2mL Representative Carton Label 1 of 2 NDC 0338-8402-02 - image 03
- Tisseel Frozen 2mL Representative Carton Label 2 of 2 NDC 0338-8402-02 - image 04
- Tisseel Lyo 2mL Representative Carton Label 1 of 2 NDC 0338-4210-02 - image 05
- Tisseel Lyo 2mL Representative Carton Label 2 of 2 NDC 0338-4210-02 - image 06
- Tisseel Lyo 2mL Representative Sleeve Label 1 of 4 NDC 0338-4301-02 - image 07
- Tisseel Lyo 2mL Representative Sleeve Label 2 of 4 NDC 0338-4301-02 - image 08
- Tisseel Lyo 2mL Representative Sleeve Label 3 of 4 NDC 0338-4301-02 - image 09
- Tisseel Lyo 2mL Representative Sleeve Label 4 of 4 NDC 0338-4301-02 - image 10
- Tisseel Frozen 2 mL - 4 mL Representative DUO Set Label - image 11
- Tisseel Lyo 2 mL - 4 mL DUPLOJET Repesentative Label 1 of 4 - image 12
- Tisseel Lyo 2 mL - 4 mL DUPLOJET Repesentative Label 2 of 4 - image 13
- Tisseel Lyo 2 mL - 4 mL DUPLOJET Repesentative Label 3 of 4 - image 14
- Tisseel Lyo 2 mL - 4 mL DUPLOJET Repesentative Label 4 of 4 - image 15
- DUPLOJET IFU Representative Label 1 of 4 - image 16
- DUPLOJET IFU Representative Label 2 of 4 - image 17
- DUPLOJET IFU Representative Label 3 of 4 - image 18
- DUPLOJET IFU Representative Label 4 of 4 - image 19
- image 20
- image 21
- image 22
- image 23
- image 24
- image 25
- image 26
- image 27
- image 28
- image 29
- image 30
- image 31
- image 32
- image 33
- image 35
- image 36
- image 37
- image 38
- image 39
Product Label Images
The following 38 images provide visual information about the product associated with Tisseel Fibrin Sealant NDC 0338-4212 by Baxter Healthcare Corporation, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Tisseel Frozen 2mL Representative Pouch Label - NDC 0338-8402-01 - image 02

Double Plunger Amber Syringe.*
Tisseel Frozen 2mL Representative Carton Label 1 of 2 NDC 0338-8402-02 - image 03

This is a description of the product Fibrin Sealant by Baxter which has an NDC code of 0338-8402-01. The product is TISSEEL which comes in a pre-filled syringe and is administered topically. It is a volatile heated, solvent and detergent treated, frozen product. The package includes Firinoysis Inhibitor (Aprotnn), Thrombin Solution (2), Thrombin (Human), and Calcium Chloride. The product should be stored at -20°C or colder and should not be exposed to temperatures above 37°C. The package includes a cautionary note to read the direction for thawing and application before use. This product is manufactured in Illinois, USA by Baxter Healthcare Corporation. The lot number and expiration date are also given.*
Tisseel Frozen 2mL Representative Carton Label 2 of 2 NDC 0338-8402-02 - image 04

This is a description of a product called Fibrin Sealant. It appears to be associated with freezing and some sort of detergent-based treatment. There are also some random numbers and words that provide no additional context about the product.*
Tisseel Lyo 2mL Representative Carton Label 1 of 2 NDC 0338-4210-02 - image 05

This is a product description for a fibrin sealant called TISSEEL, which comes in a pre-filled syringe that has been vapor heated, solvent/detergent treated, and frozen. It is indicated for topical use only and should not be injected. The product was manufactured by Baxter Healthcare Corporation and has a record number of 1501261.*
Tisseel Lyo 2mL Representative Sleeve Label 1 of 4 NDC 0338-4301-02 - image 07

Fibrin Sealant TISSEEL 2mL is a kit manufactured by Baxter Healthcare Corporation in America. The kit is Vapor Heated, Solvent/Detergent Treated, and is suitable for use in healthcare facilities to facilitate the sealing of wounds. The product is not recommended for reusing. The product holds a license in the United States under License No. 40, and it has a patent in Austria under US.Pat No: 5962405. The remaining text is not readable.*
Tisseel Lyo 2mL Representative Sleeve Label 4 of 4 NDC 0338-4301-02 - image 10

This is a description of TISSEEL 2mL, a Fibrin Sealant used as a sealer protein concentrate for human skin treatment. The contents of the package include a sealer protein concentrate, fibinolysis inhibitor, aprotin, thrombin, and gaclum chloride solution. The product comes with enclosed directions, and users are advised to discuss the risks and benefits with a doctor. It also includes the DUPLOJECT Fibin Sealant Proparation System. Note: The text has a few errors, such as "Ki" instead of "Kit," which appears to have been cut off in the scanning process.*
Tisseel Lyo 2 mL - 4 mL DUPLOJET Repesentative Label 1 of 4 - image 12
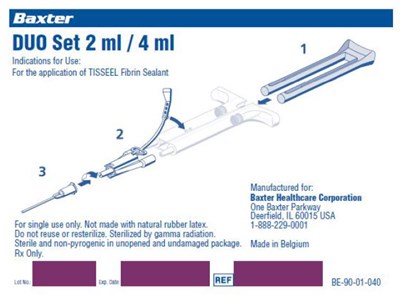
This is a set of two syringes with 2 ml and 4 ml capacity respectively, intended for the application of TISSEEL Fibrin Sealant. It is manufactured by Baxter Healthcare Corporation in Deerfield, IL and is sterile and non-pyrogenic. The product is for single use only and must not be reused or resterilized. Not-made with natural rubber atex. Sterilization is done by gamma radiation. The product is made in Belgium. No information is available if it is available only through prescription or over-the-counter.*
Tisseel Lyo 2 mL - 4 mL DUPLOJET Repesentative Label 3 of 4 - image 14

The text describes the Duploject system for preparing and applying Fibrin Sealant, which comes in a kit with either 2mL or 4 mL sizes available.*
DUPLOJET IFU Representative Label 1 of 4 - image 16

The text describes the DUPLOJECT system that can be used for preparing and applying fibrin sealant. It comes in 2mL or 4mL kits.*
DUPLOJET IFU Representative Label 2 of 4 - image 17

The BE-30-02-680 DUPLOJECT Fibrin Sealant Preparation and Application System is used for the preparation and application of TISSEEL Fibrin Sealant kit. The kit includes detailed instructions on preparation and application, and the device should be used with the FIBRINOTHERM Heating and Stirring Device. The device assists in warming the vials to the required temperature and provides instructions to ensure proper use for successful reconstitution.*
DUPLOJET IFU Representative Label 3 of 4 - image 18

Preparation instructions for the scrub nurse for the DUPLOJECT Preparation and Application System for Sealer Protein Solution and Thrombin Solution. The instructions involve opening Pack B and Packs 1, 2, and 3, assembling syringes with needles, inserting needles into the vials and withdrawing the solutions, discarding needles in sharps container, and ensuring both syringes contain the same volume.*
DUPLOJET IFU Representative Label 4 of 4 - image 19
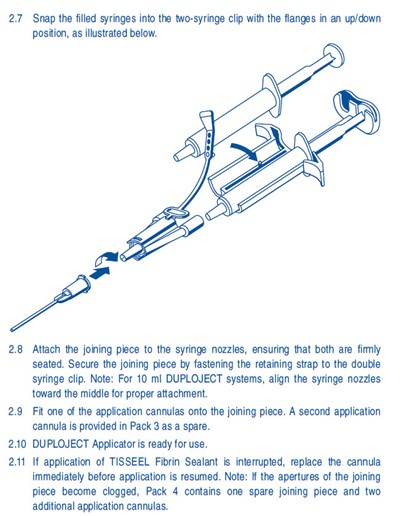
This text provides instructions on how to prepare and use the DUPLOJECT Applicator with TISSEEL Fibrin Sealant. It includes steps to snap filled syringes into a two-syringe clip, attach a joining piece to the syringe nozzles, and secure the joining piece with a retaining strap. It also directs the user to fit one of the provided application cannulas onto the joining piece and notes that a spare cannula is included in Pack 3. Finally, the text advises to replace the cannula if interrupted during application and provides information on spare parts included in Pack 4.*
image 20

This text is a warning and instruction label found on a medical product. It warns that the product is for Rx use only and should not be re-sterilized or reused. It also declares that the product is sterile, non-pyrogenic and free from natural rubber latex. The label also instructs the user to dispose of contaminated and sharp parts properly. It mentions the registered trademarks of Baxter International Inc. and provides the contact and manufacturing details for Baxter Healthcare Corporation. The revision date of the text is 2017-05-17, and the product is made in Belgium.*
image 21

This is a product description for a medical product called TISSEEL. It is a Fibrin Sealant which comes in a 2mL pre-filled syringe that is vapor heated, solvent/ detergent treated, and frozen. It is temperature sensitive and should not be exposed above 37°C (99°F). Also included is a Thrombin Solution (2) that comes in a 1mL sterile vial and contains 500 units/mL of human thrombin. The description provides lot number and expiration date details.*
image 23

Fibrin Sealant is a medical product made by Baxter. It is a topical solution that is vapor heated, solvent/detergent treated, frozen, and it comes in a pre-filled PRIMA syringe. The product should only be used topically and should not be injected. The details of the manufacturer, such as the name, address, and product number, are provided on the packaging.*
image 24

This is a product description and warning label for a Fibrin Sealant called TISSEEL. It is used topically and should not be injected and should not be exposed to temperatures above 37°C. The contents include a pre-filled syringe containing Sealer Protein Solution, Firnoge, and Thrombin Solution. The product should be stored at -20°C and should not be refrigerated or re-frozen. There are further directions for use that should be read before use. Lot number and expiration date are also provided.*
image 26

This appears to be the package information of a medical product called "TISSEEL Fibrin Sealant". It is used for topical use only and should not be injected. The product is vapor heated, solvent/detergent treated, frozen and comes with a pre-filed syringe. The package also includes order numbers and a barcode assigned by Baxter, the manufacturer.*
image 27

This is a description of a medical product called TISSEEL Fibrin Sealant. It contains 10mL of a solvent/detergent treated solution that has been frozen. The product is only for topical use and comes in a pre-filled syringe. It should not be injected or exposed to temperatures above 37°C (99°F). Instructions are provided for thawing and application before use. The product contains synthetic galcum chlorde and 3000 KIU/mL US. License No. 140. Some product details such as lot number and expiration date are missing.*
image 29

This is a description of a medical product called TISSEEL, a fibrin sealant intended for topical use only. It is supplied in a pre-filled syringe with vapor heated, solvent/detergent treated, frozen components. The manufacturer is Baxter, with locations in the USA and Australia. The product should not be injected and is only for topical use.*
image 31

This text appears to be a mix of gibberish characters and incomplete words. It is not possible to generate a useful description from it.*
image 33

Fibrin Sealant TISSEEL is a medical kit produced by Baxter Healthcare Corporation. It is a 10 mL vapor-heated, solvent/detergent treated kit, used for healthcare purposes. The product is licensed in the USA under License No. 40, and in Austria under US.Pa.No. 5062405. The additional text is not legible.*
image 38

This is a description of a medical product called "Baxter 't Fibrin Sealant TISSEEL" which is a kit with a 4ml quantity of the solution. The product is treated with vapor heating, solvent, and detergent. The manufacturer is Baxter and there is an address given in the US. In addition, there is a mention of "Bax, Duploctar Tioela v marsf Baer nb ol us Pa. o" which is not readable and unable to provide a useful description.*
image 39

Baxter's Tisseel is a kit containing 4ml of vapor heated, solvent/detergent treated Sealr protein concentrate (human), which can be used with fibrin sealant. The kit also contains 2mL of saline solution and 300K of aptin, but the product is not for injection. The risks and benefits should be discussed before use. The kit is compatible with thrombin solution. The text is difficult to read due to errors.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.














