Product Images Doxorubicin Hydrochloride, Liposomal
View Photos of Packaging, Labels & Appearance
- 10ml-carton-label - 10ml carton label
- 10ml-container-label - 10ml container label
- dhcp-page-01-of-08 - Page 01 of 08
- dhcp-page-02-of-08 - Page 02 of 08
- dhcp-page-03-of-08 - Page 03 of 08
- dhcp-page-04-of-08 - Page 04 of 08
- dhcp-page-05-of-08 - Page 05 of 08
- dhcp-page-06-of-08 - Page 06 of 08
- dhcp-page-07-of-08 - Page 07 of 08
- dhcp-page-08-of-08 - Page 08 of 08
Product Label Images
The following 10 images provide visual information about the product associated with Doxorubicin Hydrochloride, Liposomal NDC 0338-9581 by Baxter Healthcare Corporation, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10ml-carton-label - 10ml carton label
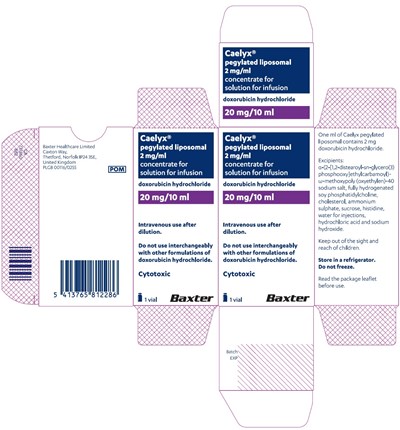
Caelyx® is a pegylated liposomal concentrate for a solution for infusion containing 2mg/ml of doxorubicin hydrochloride. It is used intravenously and should not be used interchangeably with other formulations of doxorubicin hydrochloride. It should be stored in a refrigerator and not be frozen. Caelyx® is a cytotoxic medication and should be kept out of the reach of children. Prior to use, it is important to read the package leaflet. This product is manufactured by Baxter Healthcare Limited in Thetford, Norfolk, United Kingdom.*
dhcp-page-01-of-08 - Page 01 of 08
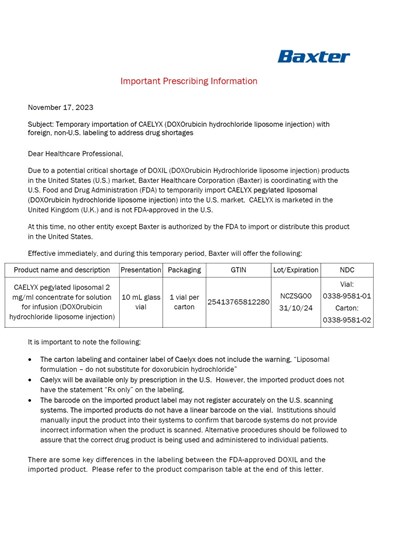
Temporary importation of CAELYX (DOXOrubicin hydrochloride liposome injection) is being coordinated by Baxter Healthcare Corporation to address a potential shortage of DOXIL (DOXOrubicin Hydrochloride liposome injection) in the United States market. CAELYX is not FDA-approved in the US but is marketed in the United Kingdom. During this temporary period, Baxter will offer CAELYX in a specific presentation and packaging. It is important to note that the labeling for CAELYX does not include a specific warning and the imported product may not register accurately on US scanning systems. Differences in labeling between DOXIL and CAELYX are outlined in a product comparison table.*
dhcp-page-02-of-08 - Page 02 of 08

Please refer to the FDA-approved package insert for DOXIL's full prescribing information at DailyMed. If you have any adverse events or product quality issues, please contact Baxter Product Surveillance at 1-800-437-5176. For adverse events associated with imported products, please call Baxter at 1-866-888-2472 or fax at 1-800-759-1801. You can also report adverse events to the FDA's MedWatch Adverse Event Reporting program online, by mail or fax. To place an order, contact Baxter's Center for Service at 1-888-229-0001. If you have any questions, contact Baxter's Medical Information Service at 1-800-933-0303.*
dhcp-page-03-of-08 - Page 03 of 08
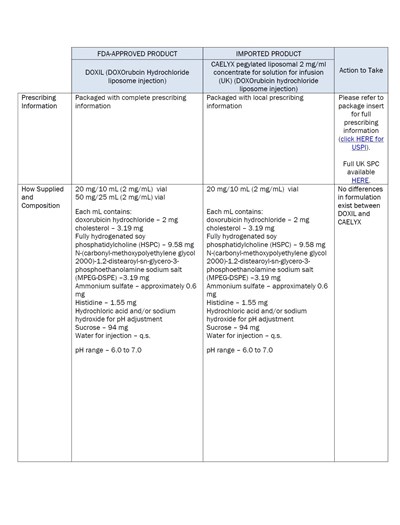
This is information about a FDA-approved product called DOXIL (DOXOrubcin Hydrochloride liposome injection) and its imported version CAELYX (pegylated liposomal 2 mg/ml concentrate for solution for infusion). The text includes details about the prescribing information, packaging, composition, and how the product is supplied. It also mentions that there are no differences in formulation between DOXIL and CAELYX.*
dhcp-page-04-of-08 - Page 04 of 08
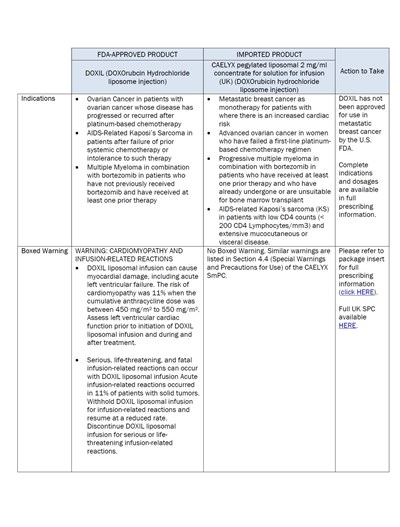
The text provided is a description and information about a FDA-approved product called DOXIL (DOXOrubcin Hydrochloride liposome injection) and its imported version called CAELYX pegylated liposomal 2 mg/ml concentrate for solution for infusion. It states the indications for the use of these products, which include ovarian cancer, AIDS-related Kaposi's Sarcoma, multiple myeloma, and metastatic breast cancer. It also mentions the warnings and precautions associated with the use of these products, such as cardiomyopathy and infusion-related reactions. The full prescribing information can be found in the package insert.*
dhcp-page-06-of-08 - Page 06 of 08
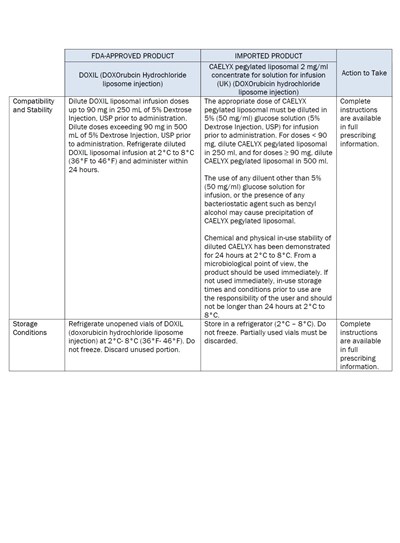
This text provides information about two FDA-approved products: DOXIL (DOXOrubicin Hydrochloride liposome injection) and CAELYX pegylated liposomal concentrate for solution for infusion. The text includes instructions for diluting and administering the products, as well as information about their compatibility and stability. It also provides storage conditions for both products.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



