Product Images Clonidine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Clonidine NDC 0378-0871 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Clonidine Cross Section - 017e8de4 474d 42cb b8ef a6b1577ae995 01
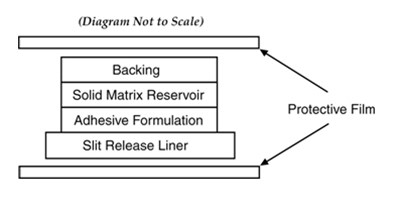
This diagram shows a solid matrix reservoir with an adhesive formulation and slit release liner. There is also a protective film included. The diagram explicitly states that it is not to scale.*
Patient Instructions Figure 2 - 017e8de4 474d 42cb b8ef a6b1577ae995 04

This text is a warning label for a medical patch that does not contain active medication. It advises the user to apply the patch and cover it with the adhesive cover if necessary. It also mentions that there are patient instructions for use. The patch contains medication and a cover, which is provided in case the patch becomes loose.*
Patient Instructions Figure 6 - 017e8de4 474d 42cb b8ef a6b1577ae995 08

This text appears to be a warning about a product called "conon1 oMESWECOVER". The warning states that the product does not contain active medication.*
image 01

This is a description of Clonidine Transdermal System, which is a type of skin patch used to deliver a medicine called Clonidine into the body over a week. Each system delivers 0.1mg Clonidine per day and contains 2.52mg Clonidine USP. It should only be used for transdermal purposes and kept out of reach of children. Dosage information can be found in the package insert, and the patch should be stored at 20-25°C. Lot and expiration information can also be recorded on the flap of the package.*
image 03

This is a description of the Clonidine Transdermal System, a medication used for transdermal delivery of 0.3 mg clonidine per day over one week. The system contains 7.56 g of clonidine, and the inactive components are colloidal silicon dioxide, mineral oil, pigmented polyethylene/polyester film, polyisobutylene, and silicone/polyester film. It is programmed for in vivo delivery and should only be used transdermally. Dosage information is included in the package insert, and the system should be stored between 20° to 25°C (68" to 77°F). The system can be found under the name Clonidine Transdermal System, USP (ECERTRY) or its corresponding NDC 0378-0873-99. The product should be kept out of reach of children.*
image 04
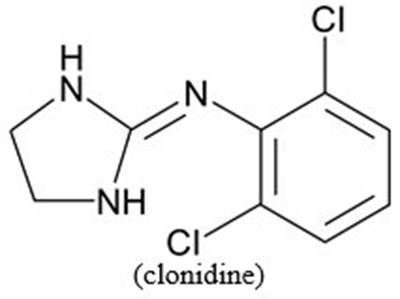
This appears to be a chemical formula for Clonidine, a medication used to treat high blood pressure and attention deficit hyperactivity disorder (ADHD).*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




