Product Images Telmisartan And Amlodipine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Telmisartan And Amlodipine NDC 0378-1078 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1a: Probability of Achieving Systolic Blood Pressure < 140 mmHg at Week 8 - image 01
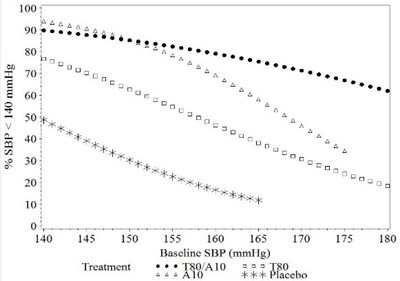
This appears to be a chart displaying blood pressure readings (in mmHg) ranging from 140 to 175 for baseline and treatment groups. The text at the top indicates that the target systolic blood pressure (SBP) is less than 140 mmHg, and there is a group receiving placebo. No further information is available from this text.*
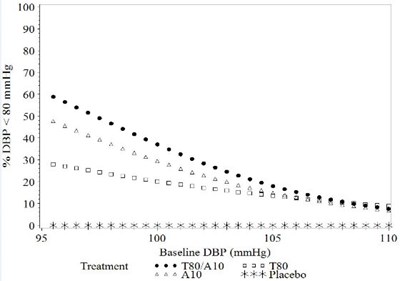
Figure 2a: Probability of Achieving Diastolic Blood Pressure < 90 mmHg at Week 8 - image 03

The text does not provide enough information to generate a useful description. Please provide more text if available.*
Telmisartan and Amlodipine Tablets 40 mg/5 mg Bottle Label - image 05
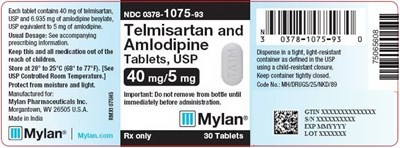
This is a description of a medication that contains both USP and amlodipine besylate. The tablets come in a container that is designed to keep them moisture and light-resistant and must be kept away from children. The prescribing information should be strictly followed, and the medication should not be removed from the bottle until just before administration. The manufacturer is Mylan Pharmaceuticals, and the tablets are intended to treat high blood pressure.*
Telmisartan and Amlodipine Tablets 40 mg/10 mg Bottle Label - image 06

This appears to be a section of a label for a medication that contains 10 mg of amlodipine. The tablets may also contain 40 mg of another substance, which is not specified. The label includes safety information, such as keeping the medication out of reach of children and storing it at a certain temperature range. There is also a warning not to remove the medication from the packaging until immediately before its administration. The name Mylan appears to be associated with the medication.*
Telmisartan and Amlodipine Tablets 80 mg/5 mg Bottle Label - image 07
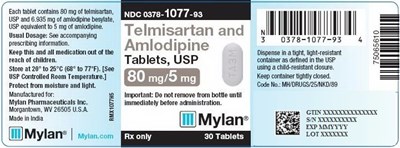
This is a medication containing 80mg of timsartan and 6595 g of amlodipine besylte, equivalent to 5 g of anlodipine. It is recommended for use with prescriptions. The medication should be kept out of the reach of children, stored between 20°C to 25°C, protected from light and moisture. The medication should not be removed from the bottle until it is immediately before administration. It is manufactured for Mylan Pharmaceuticals Inc. and made in India. There is a National Drug Code (NDC) 0378-1077-93, and a code for MYDRUGS/25NKD/SS.*
Telmisartan and Amlodipine Tablets 80 mg/10 mg Bottle Label - image 08
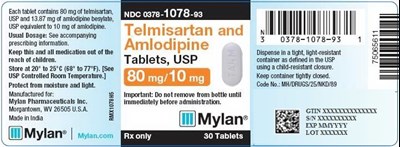
This text appears to be prescribing information for medication containing amlodipine, which is used to treat high blood pressure and angina. The medication is stored at room temperature and should be kept in a tightly closed container to protect it from moisture and light. The dosage information is not clear from this text.*
Amlodipine Besylate Structural Formula - image 09
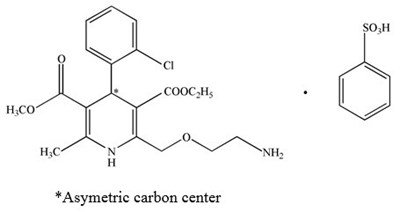
HiCO appears to be a chemical compound with an asymmetric carbon center and a possible sulfur atom denoted as "SosH". No further contextual information is provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.