Product Images Buspirone Hydrochloride
View Photos of Packaging, Labels & Appearance
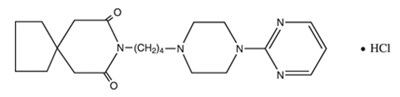
- Structural Formula - 103f116c e4f4 47d7 82aa 59a2962e465e 01
- 15 mg (the entire tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 02
- 10 mg (two-thirds of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 03
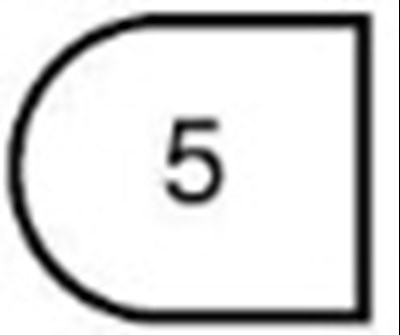
- 5 mg (one-third of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 04
- 7.5 mg (one-half of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 05
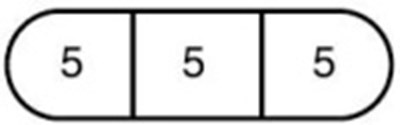
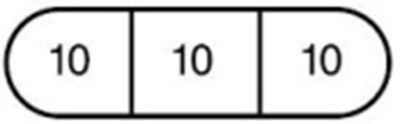
- 30 mg (the entire tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 06
- 20 mg (two-thirds of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 07
- 10 mg (one-third of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 08
- 15 mg (one-half of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 09
- Breaking Multi-Scored Tablet - image 01
- image 02
- image 03
- image 04
- image 05
- image 06
Product Label Images
The following 15 images provide visual information about the product associated with Buspirone Hydrochloride NDC 0378-1140 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image 02

Product A is a medication with 5mg of Buspirone Hydrochloride in a container with National Drug Code (NDC) 0378-1140-01 and labeled with USP. No further information is given. Product B is a container with 100 tablets, labeled with the name of the medication, BEPE, which contains 5543 Tablets of USP. The manufacturer is Adgg yan Pharmacouticals. There is a reference to Mylan®, but it is unclear if it is related to the product B or not.*
image 05

This is a medication description for Buspirone Hydrochloride tablets in a strength of 15mg. The tablets come in a bottle with NDC number 0378-1165-91, and the recommended storage temperature is between 20° to 25°C. The tablets are to be dispensed only with a prescription.*
image 06

This is a label for medication, called Cpua Tablets, USP. It advises to store the medication between 20° to 25°C (68° to 77°F) and to keep the container tightly closed. It also mentions the manufacturer, Mylan, and that it is a prescription medication.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.