Product Images Amlodipine And Valsartan
View Photos of Packaging, Labels & Appearance
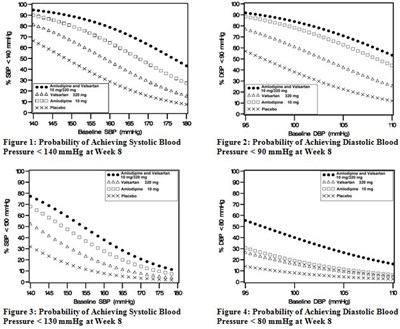
- Amlodipine and Valsartan Figures 01 - 04 - image 01
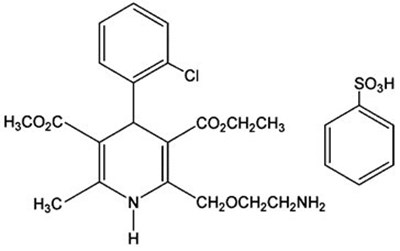
- Amlodipine Besylate Structural Formula - image 02
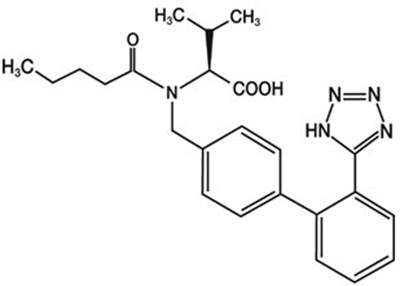
- Valsartan Structural Formula - image 03
- Amlodipine and Valsartan Tablets, USP 5 mg/160 mg Bottle Label - image 04
- Amlodipine and Valsartan Tablets, USP 10 mg/160 mg Bottle Label - image 05
- Amlodipine and Valsartan Tablets, USP 5 mg/320 mg Bottle Label - image 06
- Amlodipine and Valsartan Tablets, USP 10 mg/320 mg Bottle Label - image 07
Product Label Images
The following 7 images provide visual information about the product associated with Amlodipine And Valsartan NDC 0378-1724 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Amlodipine and Valsartan Tablets, USP 5 mg/160 mg Bottle Label - image 04

Each film-coated tablet contains 6.94 mg of Amlodipine Besylate, USP (equivalent to 5 mg of Amlodipine) and 160 mg of Valsartan, USP. The usual dosage is indicated in the accompanying prescribing information. This medication should be kept out of the reach of children and stored at a controlled room temperature of 20° to 25°C (63° to 77°F). Protect from moisture. The drug is manufactured for Mylan Pharmaceuticals Inc. and is not available in India. The product has an NDC code 0378-1721-93 and is dispensed in a high-resistant container as defined in the USP, using a high-resistant closure. The container should be kept tightly closed, and a prompt code will be printed along with variable data online coding.*
Amlodipine and Valsartan Tablets, USP 10 mg/160 mg Bottle Label - image 05

This is a medication containing 13.88 mg of amlodipine besylate, USP, equivalent to 10 mg of amlodipine. The medication is a film-coated tablet produced by Mylan Pharmaceuticals. Each tablet has the NDC code 0378-1722-03 and should be stored at a temperature ranging from 20°C to 26°C. The medication is child-resistant and should be kept out of reach. There are additional details about the manufacturer, origin, and variable data coding on the packaging.*
Amlodipine and Valsartan Tablets, USP 5 mg/320 mg Bottle Label - image 06

This is a product description of Amlodipine Besylate tablets manufactured by Mylan Pharmaceuticals. Each film-coated tablet contains 6.94 mg of amlodipine besylate, USP. The tablets are stored at controlled room temperature and must be protected from moisture. They come in a container with a chill-resistant closure and are dispensed in a tight, light-resistant container as defined in the USP. The description also includes the manufacturer's address, product code, and lot and expiry dates.*
Amlodipine and Valsartan Tablets, USP 10 mg/320 mg Bottle Label - image 07

This is a description of Amlodipine and Valsartan Tablets, which are film-coated tablets that contain 13.88 mg of amlodipine besylate (equivalent to 10mg of amlodipine) and 320 mg of valsartan. The prescribed dosage is not provided in this text and should be checked in the prescribing information. The medication should be kept out of the reach of children, stored at a temperature of 20°C to 26°C (63°F to 77°F) and protected from moisture. The tablets are manufactured by Mylan Pharmaceuticals Inc. and dispensed in a tightly closed, light-resistant container as defined by the United States Pharmacopeia (USP). The text also provides information regarding the product's size, NDC code, and manufacturing country, which is India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.