Product Images Amlodipine And Atorvastatin
View Photos of Packaging, Labels & Appearance
- Amlodipine Besylate and Atorvastatin Calcium Structural Formulae - image 01
- Figure 1. Kaplan-Meier Analysis of Composite Clinical Outcomes for Amlodipine vs. Placebo - image 02
- Figure 2. Effects on Primary Endpoint of Amlodipine vs. Placebo across Sub-Groups - image 03
- Figure 3. Effect of Atorvastatin 10 mg/day on Cumulative Incidence of Non-Fatal Myocardial Infarction or Coronary Heart Disease Death (in ASCOT-LLA) - image 04
- Figure 4. Effect of Atorvastatin 10 mg/day on Time to Occurrence of Major Cardiovascular Events (Myocardial Infarction, Acute CHD Death, Unstable Angina, Coronary Revascularization, or Stroke) in CARDS - image 05
- Figure 5. Effect of Atorvastatin 80 mg/day vs. 10 mg/day on Time to Occurrence of Major Cardiovascular Events (TNT) - image 06
- Amlodipine Besylate and Atorvastatin Calcium Tablets 2.5 mg/10 mg Bottle Label - image 07
- Amlodipine Besylate and Atorvastatin Calcium Tablets 2.5 mg/20 mg Bottle Label - image 08
- Amlodipine Besylate and Atorvastatin Calcium Tablets 2.5 mg/40 mg Bottle Label - image 09
- Amlodipine Besylate and Atorvastatin Calcium Tablets 5 mg/10 mg Bottle Label - image 10
- Amlodipine Besylate and Atorvastatin Calcium Tablets 5 mg/20 mg Bottle Label - image 11
- Amlodipine Besylate and Atorvastatin Calcium Tablets 5 mg/40 mg Bottle Label - image 12
- Amlodipine Besylate and Atorvastatin Calcium Tablets 5 mg/80 mg Bottle Label - image 13
- Amlodipine Besylate and Atorvastatin Calcium Tablets 10 mg/10 mg Bottle Label - image 14
Product Label Images
The following 14 images provide visual information about the product associated with Amlodipine And Atorvastatin NDC 0378-4514 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Amlodipine Besylate and Atorvastatin Calcium Structural Formulae - image 01
This is a list of some chemical names, it doesn't provide a useful description.*
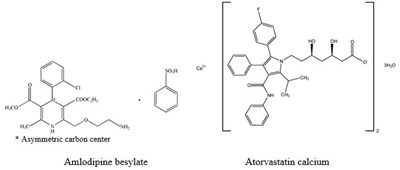
Figure 1. Kaplan-Meier Analysis of Composite Clinical Outcomes for Amlodipine vs. Placebo - image 02
This appears to be output from a statistical analysis of some kind, likely related to a medical study involving a medication called "Amlodipine". The "Event Rate" is listed at 0.0 and there is a P-value of 0.003 and Hazard Ratio of 0.691, along with a 95% CI between 0.54 and 0.88. There are also some numerical values listed with the heading "at risk" and a time frame indicated in months. The specifics of the analysis and their implications are not clear without additional context.*
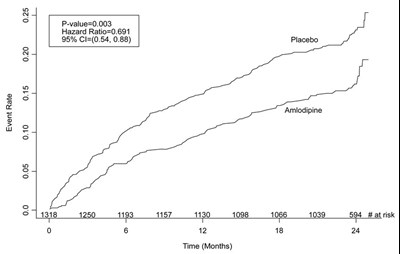
Figure 2. Effects on Primary Endpoint of Amlodipine vs. Placebo across Sub-Groups - image 03
This is a table showing the distribution of patients (male and female) based on various factors such as baseline systolic blood pressure, vessel disease, and stenosis. The table also includes the number of patients who underwent PCI (percutaneous coronary intervention) with or without stent placement. The mean sitting baseline SBP is 129 mmHg.*
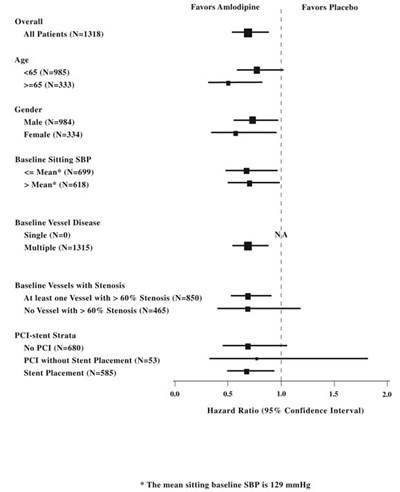
Figure 3. Effect of Atorvastatin 10 mg/day on Cumulative Incidence of Non-Fatal Myocardial Infarction or Coronary Heart Disease Death (in ASCOT-LLA) - image 04
The text represents a graph with cumulative incidence percentages for Atorvastatin and Placebo over a period of 35 years. The HR (hazard ratio) with its corresponding confidence interval and p-value is also mentioned.*
Figure 4. Effect of Atorvastatin 10 mg/day on Time to Occurrence of Major Cardiovascular Events (Myocardial Infarction, Acute CHD Death, Unstable Angina, Coronary Revascularization, or Stroke) in CARDS - image 05
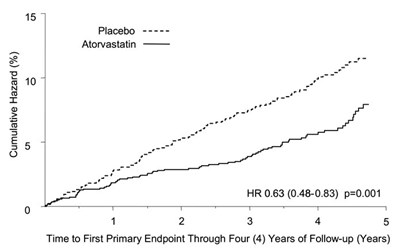
The text describes a cumulative hazard graph that shows data for a placebo and a medication called Atorvastatin. There is also information on hazard ratio (HR) and confidence intervals (0.48-0.83) for the medication. The x-axis shows time to the first primary endpoint through four years of follow up.*
Figure 5. Effect of Atorvastatin 80 mg/day vs. 10 mg/day on Time to Occurrence of Major Cardiovascular Events (TNT) - image 06
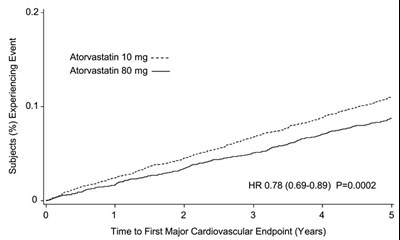
This is a description of a clinical trial that evaluated the effects of Atorvastatin, a medication used to lower cholesterol levels, on major cardiovascular events. The table shows the percentage of subjects who experienced the event, and the graph displays the time to the first major cardiovascular endpoint in years. The study found that Atorvastatin at a dosage of 80mg reduced the risk of the event by 22% (HR 0.78, CI 0.69-0.89, P=0.0002) compared to a lower dosage of 10mg.*
Amlodipine Besylate and Atorvastatin Calcium Tablets 2.5 mg/10 mg Bottle Label - image 07

This is a description of a medication called Amlodipine and Atorvastatin, which is sold in the form of tablets. The tablets are produced by Viron Pharmaceuticals and have a unique NDC identification code of 0378-4513-93. The tablets are marked with the brand name Mylan and are available by prescription only. The medication is used to treat high blood pressure and high cholesterol and comes in a 30-tablet pack. The packaging includes a prompt to print the lot number, expiry date, and variable data online coding for each tablet.*
Amlodipine Besylate and Atorvastatin Calcium Tablets 2.5 mg/40 mg Bottle Label - image 09
Each tablet contains 1543375 mg of calcium and is a controlled temperature drug, manufactured by Mylan Pharmaceuticals. It is a USP tablet and dosage instructions are included. The text also includes lot and expiry information along with a coding reference for variable data online.*
Amlodipine Besylate and Atorvastatin Calcium Tablets 5 mg/10 mg Bottle Label - image 10

This is a medication called Amlodipine and Atorvastatin Tablets, USP. Each coated tablet contains 5 mg of Amlodipine equivalent to 6.9 mg Amlodipine and 1986751 mg Atorvastatin. The recommended dosage should be obtained from the prescribing information. It should be stored at controlled room temperature between 20-25°C (68-77°F). This medication is manufactured by Mylan Pharmaceuticals Inc. in India. The packaging contains 30 tablets and the NDC number is 0378-4516-93. It must be kept out of reach of children and kept in a tightly sealed, light-resistant container. The code number is MHDRUGS25MKD/B and there is some variable data coding online available. The LOT and EXP information would be printed along with other variable data, whenever applicable.*
Amlodipine Besylate and Atorvastatin Calcium Tablets 5 mg/20 mg Bottle Label - image 11

This appears to be a medication label containing information including the drug name, dosage, manufacturer name and identification numbers for the drug. The label also includes a warning not to use the medication outside of a doctor's recommendation and a section for variable data coding.*
Amlodipine Besylate and Atorvastatin Calcium Tablets 5 mg/40 mg Bottle Label - image 12

This appears to be a pharmaceutical packaging label with information such as a phone number and a product name "Ao Tablets, USP". The label also includes a prompt for printing "LOT" and "EXP" for variable data coding. There is a code number "1234567" printed at the end, along with some other numbers and symbols.*
Amlodipine Besylate and Atorvastatin Calcium Tablets 5 mg/80 mg Bottle Label - image 13

This seems to be a label of a medication called Amlodipine and Atorvastatin, in tablet form. The label also includes a code for identification (LOT) and an expiration date (EXP) for quality control. There is also a mention of a variable data online coding system.*
Amlodipine Besylate and Atorvastatin Calcium Tablets 10 mg/10 mg Bottle Label - image 14

This is a description of a medication called Amlodipine and Atorvastatin Tablets, USP. Each tablet contains 10mg of Amlodipine equivalent to 13.83mg of Amlodipine besylate, USP, and 80mg of Atorvastatin equivalent to 86.751mg of Atorvastatin calcium, USP. The usual dosage is not provided in the text and must be obtained from accompanying prescribing information. The medication should be kept out of reach of children and stored at 20-25°C. The tablets are manufactured by Myl Pharmaceuticals Inc. and are dispensed in a light-resistant container. The text also includes a product code, but the information on how to use it is not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
