Product Images Prasugrel
View Photos of Packaging, Labels & Appearance
- Figure 1: Non-CABG-Related TIMI Major or Minor Bleeding Events. - image 01
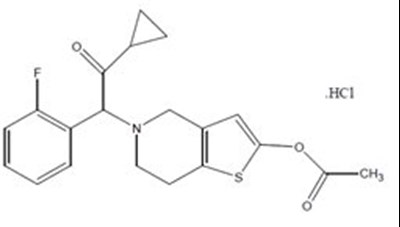
- Prasugrel Hydrochloride Structural Formula - image 02
- Figure 2: Inhibition (Mean ± SD) of 20 μM ADP-induced Platelet Aggregation (IPA) Measured by Light Transmission Aggregometry after Prasugrel 60 mg. - image 03
- Figure 3: Time to first event of CV death, MI, or stroke (TRITON-TIMI 38). - image 04
- Figure 4: Subgroup analyses for time to first event of CV death, MI, or stroke (HR and 95% CI; TRITON-TIMI 38) – UA/NSTEMI Patients. - image 05
- Figure 5: Subgroup analyses for time to first event of CV death, MI, or stroke (HR and 95% CI; TRITON-TIMI 38) – STEMI Patients. - image 06
- Prasugrel Tablets 5 mg Bottle Label - image 07
- Prasugrel Tablets 10 mg Bottle Label - image 08
Product Label Images
The following 8 images provide visual information about the product associated with Prasugrel NDC 0378-5186 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2: Inhibition (Mean ± SD) of 20 μM ADP-induced Platelet Aggregation (IPA) Measured by Light Transmission Aggregometry after Prasugrel 60 mg. - image 03

Figure 3: Time to first event of CV death, MI, or stroke (TRITON-TIMI 38). - image 04

The given text seems to be a graph or a figure, with some text scattered around it. The text is not coherent and is incomplete. Therefore, the description is not available.*
Figure 4: Subgroup analyses for time to first event of CV death, MI, or stroke (HR and 95% CI; TRITON-TIMI 38) – UA/NSTEMI Patients. - image 05

Figure 5: Subgroup analyses for time to first event of CV death, MI, or stroke (HR and 95% CI; TRITON-TIMI 38) – STEMI Patients. - image 06

Prasugrel Tablets 5 mg Bottle Label - image 07

This is a medication containing 5 mg of prasugrel hydrochloride, USP. The usual dosage should be consulted using the accompanying prescribing information. It should be kept away from children and in a controlled room temperature within the original container with the desiccant. The dispenser should provide a medication guide for each patient. There are 30 tablets in the container. The inner seal must not be used if missing or broken. Finally, there is a LOT number and a serial number on the container.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.