Product Images Levetiracetam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Levetiracetam NDC 0378-5615 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
export 00

This is a medication bottle label with instructions on how to store and dispense tablets. It includes information on the temperature range for storage and the use of child-resistant closures. It also details the drug code and advises pharmacists to provide a medication guide to each patient. It is manufactured by Hylan Pharmaceuticals Inc. However, the text appears to be incomplete and fragmented, making it hard to discern the complete instructions.*
export 01
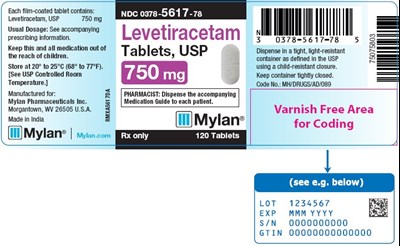
This text appears to be a description of the storage and packaging instructions for the medication called Levetiracetam. The medication comes in tablet form with a strength of 750 mg. It should be stored at a temperature between 20°C and 25°C and kept in a tightly closed container with a child-resistant closure. There is a reference to a USP Controlled Room Temperature code. The manufacturer is identified as Hyin Pharmaceuticals, and the medication guide should be dispensed with the tablets. However, the text also includes various random characters and incomplete words, possibly due to errors.*
export 02
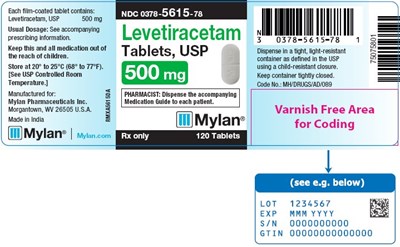
This is a description of a medication bottle label for a drug called Levetiracetam, which is in the form of tablets. The label includes instructions for storing the medication at a specific temperature range and dispensing it in a tight container with a child-resistant closure. The label also includes a product code and manufacturer information, including the company name and location. There is also a date code on the label.*
export 03
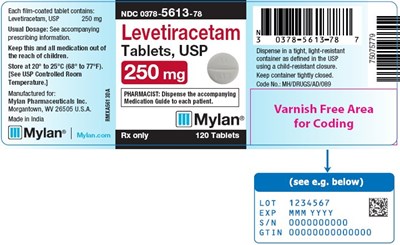
Each film-coated tablet contains A nootropic drug called Levetiracetam USP. The tablets come in a container and should be stored at a temperature between 20°C to 25°C. The container has a child-resistant closure. The manufacturer S Hyin Pharmaceuticals provides 120 tablets in one container. The rest of the text is not available or recognizable.*
Figure 5: Responder Rate for All Patients Ages 1 Month to < 4 Years (≥ 50% Reduction from Baseline) in Study 5 - image 06
This text provides a formula to calculate the total daily dose of a medication based on the patient's weight and the daily dose of the medication in milligrams per kilogram (mg/kg). The medication concentration is given as 100 mg/mL.*
Figure 6: Responder Rate (≥ 50% Reduction from Baseline) in PGTC Seizure Frequency per Week in Study 7 - image 07
This text likely represents a formula for calculating creatinine clearance (CLer) in patients. The formula requires the patient's age in years and weight in kilograms, with the result of 140 minus the age multiplied by the weight. For female patients, this result should be multiplied by 0.85. Additionally, the formula requires the patient's serum creatinine level in milligrams per deciliter (mg/dL), which should be multiplied by 72.*
Levetiracetam Tablets, USP 500 mg Bottle Label - image 09
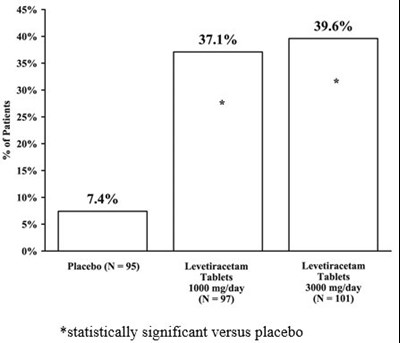
The given text represents a statistical data of some experiment which might compare the effectiveness of placebo with Levetiracetam tablets. It shows the percentages of different values such as 5%, 39.6%, 37.1%, 30%, 0%, 15%, 10%, and 7.4%. The asterisk mark (*) beside the percentage of 30 indicates its statistical significance versus placebo. As there is no clear context provided, it is not possible to ascertain the exact nature or purpose of the experiment or the significance of these percentages.*
Levetiracetam Tablets, USP 750 mg Bottle Label - image 10
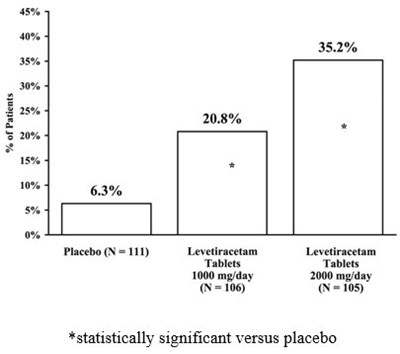
This text appears to be a chart showing percentages for some variables, including a comparison between a placebo and a medication called Levetiracetam at two different dosages. The medication had a statistically significant effect compared to the placebo, but without more information, it's not clear what the variables being measured were or what the context of the study was.*
image 12
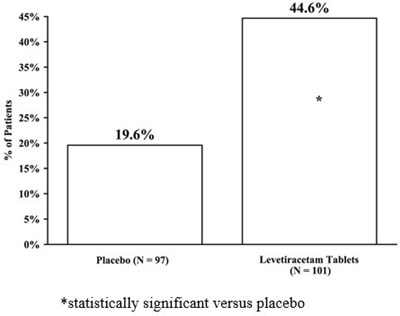
The text provides the percentage of patients for a medical treatment, with 44.6% being the percentage achieved by Placebo and 19.6% being the percentage achieved by Levetiracetam Tablets. The text also mentions that the efficacy of Levetiracetam Tablets was statistically significant when compared to Placebo.*
image 13
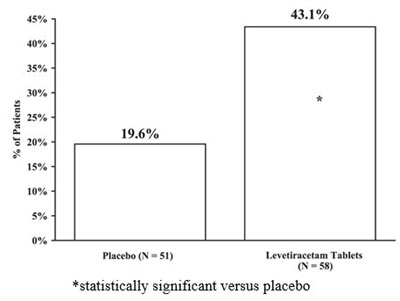
The text represents a statistical report of a medical trial. The trial involved two groups of patients, one treated with Levetiracetam Tablets and another administered Placebo. The report provides the percentage of patients from each group and indicates a statistically significant difference in the outcomes between the two groups indicating the effectiveness of the medication.*
image 14
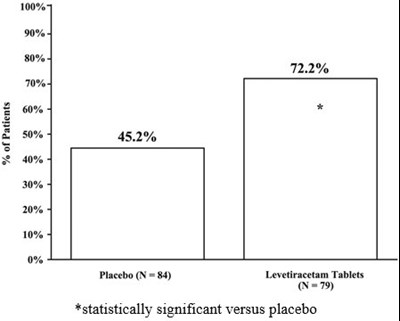
This text provides some numerical values and mentions a placebo group in a study involving Levetiracetam tablets. The percentage values such as "w0%", "722%", "H 45.2%", and "2 0%" are not explained or given a context, making it difficult to interpret their meaning. The text also suggests that the study found a statistically significant difference between the treatment group and the placebo group, but it is unclear what the treatment group was or what outcome measure was used. Overall, the provided text is not clear and informative enough to generate a useful description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


