Product Images Norethindrone Acetate And Ethinyl Estradiol
View Photos of Packaging, Labels & Appearance
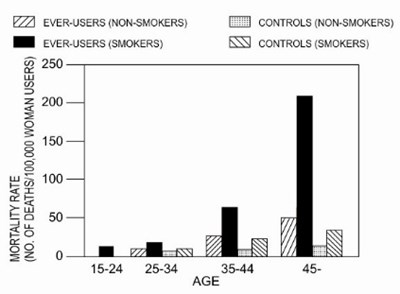
- Graph - 6ba530ce 2499 4963 850e 620b8956af7b 02
- Blister image with instructions - 6ba530ce 2499 4963 850e 620b8956af7b 03
- Day Label Sticker - 6ba530ce 2499 4963 850e 620b8956af7b 04
- Blister image with instructions - 6ba530ce 2499 4963 850e 620b8956af7b 05
- Day Sticker Label - 6ba530ce 2499 4963 850e 620b8956af7b 06
- image 01
- image 02
- image 03
Product Label Images
The following 8 images provide visual information about the product associated with Norethindrone Acetate And Ethinyl Estradiol NDC 0378-7283 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Blister image with instructions - 6ba530ce 2499 4963 850e 620b8956af7b 03

This text provides instructions to start taking birth control pills. It includes guidelines for both Sunday and Day-1 starters and instructions for placing the day label strip. The text also mentions the name, dosage, and storage temperature of the birth control pills. It advises taking the pills from left to right each week.*
Day Label Sticker - 6ba530ce 2499 4963 850e 620b8956af7b 04

This text appears to be a set of instructions for using a period tracker. It instructs the user to select the day label strip that corresponds with the start of their period and place it over a pre-printed area with the days of the week. The text includes abbreviations for the days of the week, and mentions skipping steps if the first day of the period falls on a Sunday.*
Blister image with instructions - 6ba530ce 2499 4963 850e 620b8956af7b 05

This is a set of instructions for people on oral contraceptive medication. The text provides guidance on how to begin the medication pack, depending on the start date of the menstrual cycle. The medication is described as Norethindrone Acetate and EthinylEstradiol Tablots USP, 1 mg/0.02 mg and Forrous Fumarato Tablots. The user is instructed to take the pills from left to right each week and store them at room temperature.*
Day Sticker Label - 6ba530ce 2499 4963 850e 620b8956af7b 06

This text appears to be a set of instructions for using a menstrual cycle tracking tool. It provides guidance on how to select and apply a day label strip to mark the start of a menstrual cycle on a preprinted calendar that displays the days of the week. The text also notes that if the first day of a menstrual period starts on a Sunday, the aforementioned step can be skipped.*
image 01

Norethindrone Acetate and Ethinyl Estradiol Tablets, USP and Ferrous Fumarate Tablets are intended for use as oral contraceptives to prevent pregnancy. The product contains 21 tablets in a pack, with each tablet containing 1mg norathindrone acetaa and 0.02mg etinyl asradiol. The pack also includes 4 brown tablets. This product does not protect against sexually transmitted diseases, including AIDS. The Patient Instructions included in each pouch provide instructions to the patient. Store at 20°-25°C (68"-77°F) and keep out of the reach of children.*
image 02
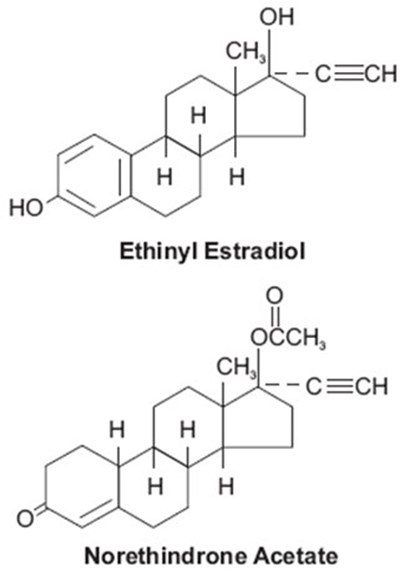
Norethindrone Acetate is a medication used for birth control and hormonal disorders. It is a type of progestin hormone that works by preventing ovulation and altering the cervical mucus to make it harder for sperm to reach the egg. Norethindrone Acetate is available in various forms, including pills, injections, and implants. It is important to take this medication as prescribed to ensure effectiveness and prevent any potential side effects.*
image 03
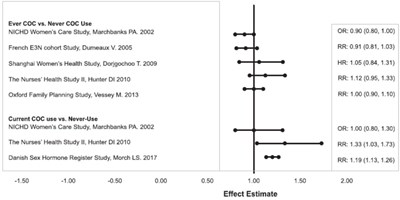
This text provides a list of studies with their respective effect estimates, comparing Ever COC (combined oral contraceptive pill) users versus Never COC users, as well as the effect estimate of Current COC use versus Never-use. The studies include the NICHD Women's Care Study, French E3N cohort Study, Shanghai Women's Health Study, The Nurses' Health Study II, and the Oxford Family Planning Study. The effect estimate is presented as OR, RR, or HR values along with their corresponding confidence intervals. It is unclear what the studies were investigating.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.