Product Images Nitroglycerin
View Photos of Packaging, Labels & Appearance
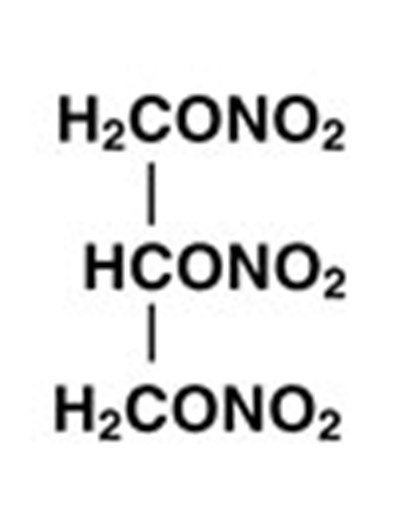
- Nitroglycerin Structural Formula - image 01
- Patch Cross Section - image 02
- Patient Information Figure 01 - image 03
- Patient Information Figure 02 - image 04
- Patient Information Figure 03 - image 05
- Patient Information Figure 04 - image 06
- Patient Information Figure 05 - image 07
- Nitroglycerin Transdermal System 0.1 mg/hr Carton - image 08
- Nitroglycerin Transdermal System 0.2 mg/hr Carton - image 09
- Nitroglycerin Transdermal System 0.4 mg/hr Carton - image 10
- Nitroglycerin Transdermal System 0.6 mg/hr Carton - image 11
Product Label Images
The following 11 images provide visual information about the product associated with Nitroglycerin NDC 0378-9116 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Patch Cross Section - image 02
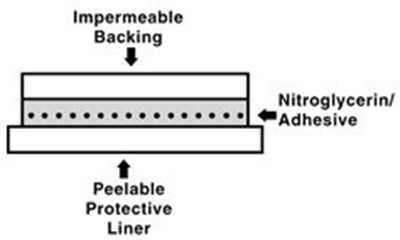
This text appears to be describing the properties of a product, likely an adhesive or coating. The product is impermeable and has a peelable protective backing. It also contains nitroglycerin, which may suggest medical or industrial applications. However, the text is incomplete and difficult to interpret without further context or information.*
Nitroglycerin Transdermal System 0.1 mg/hr Carton - image 08

This is a description of a transdermal system for the drug Nitroglycerin. The system contains 11.2mg of nitroglycerin and is for transdermal use only. The inactive components consist of acryl, polyester, silicone, and other materials. The system should be stored in a controlled room temperature. The text also includes some warnings and instructions.*
Nitroglycerin Transdermal System 0.2 mg/hr Carton - image 09

This is a description of the Nitroglycerin Transdermal System manufactured by Mylan. Each system contains 22.4 mg of nitroglycerin and various adhesive components. The product is for transdermal use only and should be applied immediately upon removal from the packaging. The text includes instructions for application and a patient should read and follow the instructions carefully. The blurred and distorted characters make it difficult to understand the last part of the text.*
Nitroglycerin Transdermal System 0.4 mg/hr Carton - image 10

This is a description of a nitroglycerin transdermal system, which is a prescription-only medication used to treat symptoms related to chest pain caused by angina. Each patch contains 44.8 mg of nitroglycerin, and the instructions for use include opening the pouch, carefully removing the patch, placing it on the chosen skin site and pressing it firmly in place, with the recommendation to not touch the exposed sticky side of the patch. The system contains inert components such as acryl pressure-sensitive adhesive, with a cross-linking agent polyolefin, polyester elastomer coated on PET film with silicones and white ink containing tartrazine. This product is for transdermal use only and the instructions have more detailed information on the back panel.*
Nitroglycerin Transdermal System 0.6 mg/hr Carton - image 11
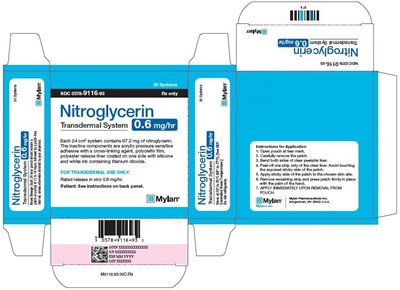
This is a description for a Nitroglycerin Transdermal System used for transdermal applications. Each 24-hour system contains 67.2 mg of nitroglycerin, and its inactive components include acrylic pressure-sensitive adhesive with a cross-linking agent, polyolefin film, polyester release liner coated on one side with silicone and white ink containing titanium dioxide. The release rate is at 0.6 ma/h. Instructions for application are also available on the packaging.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.