Product Images Fentanyl
View Photos of Packaging, Labels & Appearance
- Medication Guide Fentanyl Packaging Layers - image 01
- Medication Guide Figures A - D - image 02
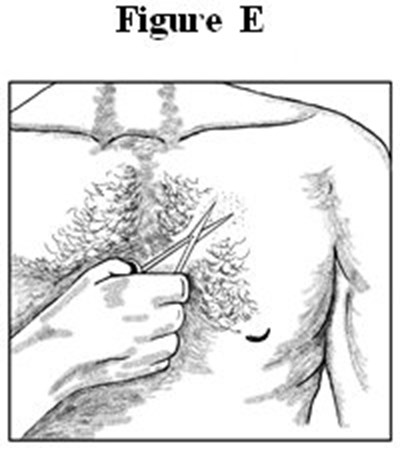
- Medication Guide Figures E - image 03
- Medication Guide Figures F - image 04
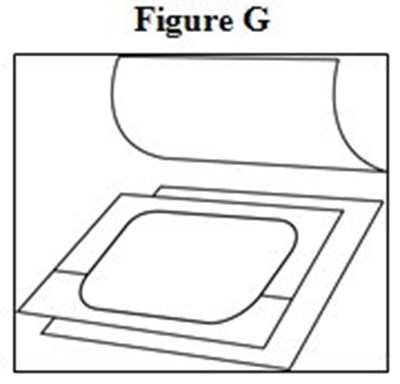
- Medication Guide Figures G - image 05
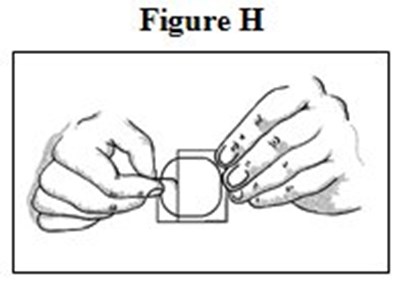
- Medication Guide Figures H - image 06
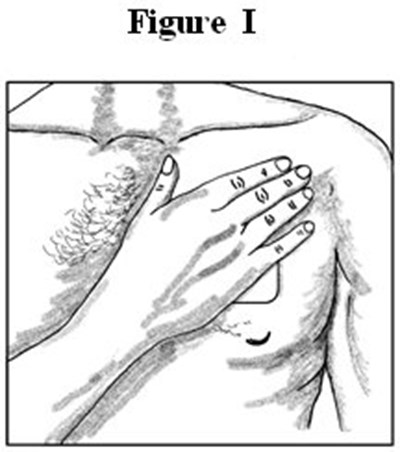
- Medication Guide Figures I - image 07
- Medication Guide Figures J - image 08
- Medication Guide Figures K - image 09
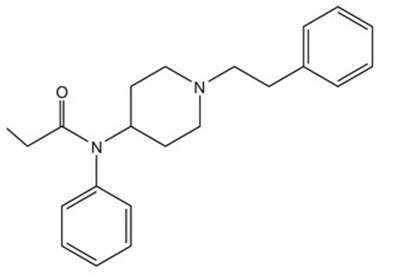
- Fentanyl Structural Formula - image 10
- Fentanyl Transdermal Protective Film - image 11
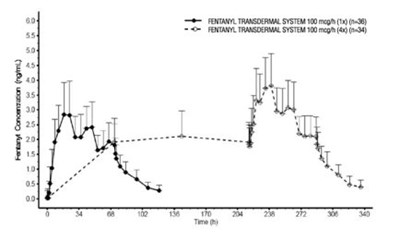
- Figure 1 Serum Fentanyl Concentrations Following Single and Multiple Applications of Fentanyl Transdermal System 100 mcg/hr - image 12
- Fentanyl Transdermal System 12 mcg/hr Carton Label - image 13
- Fentanyl Transdermal System 25 mcg/hr Carton Label - image 14
- Fentanyl Transdermal System 37.5 mcg/hr Carton Label - image 15
- Fentanyl Transdermal System 50 mcg/hr Carton Label - image 16
- Fentanyl Transdermal System 62.5 mcg/hr Carton Label - image 17
- Fentanyl Transdermal System 75 mcg/hr Carton Label - image 18
- Fentanyl Transdermal System 87.5 mcg/hr Carton Label - image 19
- Fentanyl Transdermal System 100 mcg/hr Carton Label - image 20
Product Label Images
The following 20 images provide visual information about the product associated with Fentanyl NDC 0378-9124 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Medication Guide Fentanyl Packaging Layers - image 01

The text seems to be a list of two product features: a Protective Film Backing Layer and a Release Liner. It is unclear what type of product this refers to, but it appears to be related to some sort of protective material.*
Figure 1 Serum Fentanyl Concentrations Following Single and Multiple Applications of Fentanyl Transdermal System 100 mcg/hr - image 12

This appears to be a brief description of a transdermal patch. The first layer is a protective film, followed by a backing layer. The drug is contained within a middle layer. Finally, there is a release liner that needs to be removed before applying the patch to the skin.*
Fentanyl Transdermal System 12 mcg/hr Carton Label - image 13

This appears to be a product information sheet for Fentanyl Transdermal System by Mylan Pharmaceuticals Inc. It includes information on inactive ingredients and usual dosage, as well as instructions for use and storage. It also includes important safety information, warnings, and a phone number for questions or medical emergencies. The product is a transdermal system that delivers 12 mg/hr of fentanyl for 72 hours and should not be used for certain types of pain or by those who are not opioid-tolerant. The sheet advises users to read the enclosed Fentanyl Transdermal System Modification Guide for additional safety information.*
Fentanyl Transdermal System 25 mcg/hr Carton Label - image 14

The text appears to be a description of a medication called Fentanyl Transdermal System. The medication is delivered through the skin and is used for pain management. The dosage and application instructions are provided, along with the inactive ingredients. The medication packaging has important safety information, and there is a convenient area to record narcotic use. The text also includes contact information for the pharmaceutical company and a warning not to use the medication if the seal on the pouch is broken.*
Fentanyl Transdermal System 37.5 mcg/hr Carton Label - image 15

This is a medication label describing Fentanyl Transdermal System, a patch applied to the skin that delivers 375 mcg/hr fentanyl for 72 hours. It is used for severe pain that cannot be treated with immediate-release opioids or non-opioid analgesics, and should not be used for intermittent pain. Each package contains five systems. The patch should be applied immediately after removal from the pouch, and the area should not be exposed to heat. Store in the original unopened pouch at 68 to 77°F. Read the enclosed Medication Guide before use. If the seal is broken, do not use, and keep out of reach of children. If you have questions, call Mylan Pharmaceuticals Inc. If you are experiencing a medical emergency, call 911.*
Fentanyl Transdermal System 50 mcg/hr Carton Label - image 16

Fentanyl Transdermal System is a drug delivery system that is used to deliver 50 mcg/hr of Fentanyl for 72 hours. It contains 510 mg of Fentanyl and must be applied immediately after removal from the pouch and the protective liner is removed. The area of application must not be exposed to heat and the system should be stored at a temperature of 20° to 25°C. The medication guide must be read for important safety information, and it should not be used for non-opioid analgesics or for pain that can be treated with immediate-release opioids. In case of any questions, reach Mylan Pharmaceuticals Inc. at 1-877-446-3679 (1-8774-INFO-RX).*
Fentanyl Transdermal System 62.5 mcg/hr Carton Label - image 17

This text appears to be a description of a medication called Fentanyl Transdermal System. It includes information such as the inactive ingredients, usual dosage, storage instructions, and a warning against using the medication in certain situations. There is also contact information for questions or emergencies related to the medication.*
Fentanyl Transdermal System 75 mcg/hr Carton Label - image 18

This is a product description of Fentanyl Transdermal System by Mylan Pharmaceuticals Inc., indicated for delivering 75 mcg/hr fentanyl for 72 hours for pain management in people who are opioid-tolerant. It contains inactive ingredients like Dimethicone NF, and silicone adhesive. Every transdermal system comprises 7.65 mg of fentanyl. The product should not be used for treating pain that can be relieved by immediate-release opioids or non-opioid analgesics or on an intermittent, postoperative basis unless the patient has used other narcotic opioid medicines before. It comes with an enclosed fentanyl transdermal system medication guide that provides essential safety information, and the seal on the pouch should not be broken. For more information and questions, one can call Mylan Pharmaceuticals Inc. at 1-877-446-3678.*
Fentanyl Transdermal System 87.5 mcg/hr Carton Label - image 19

This is a description for a Fentanyl Transdermal System that provides in vivo delivery of 87.5 mcg/hr fentanyl for 72 hours. The transdermal system contains 8.93 mg fentanyl and has inactive ingredients of Dimethicone NF and silicone adhesive. It is advised to apply immediately upon removal from the pouch and after removal of the protective liner. This medicine should not be used for pain that can be treated with immediate-release opioids or non-opioid analgesics, for intermittent or postoperative pain, and unless you are opioid tolerant. The medication guide should be read for important safety information. Emergency contact information and dosage information can also be found on the packaging.*
Fentanyl Transdermal System 100 mcg/hr Carton Label - image 20

The text is a description of the Fentanyl Transdermal System. It warns against using the system for non-opioid treatable pain, post-operative pain, or if you are not opioid tolerant. It also contains product information, such as dosage, ingredients, storage instructions, and contact information for Mylan Pharmaceuticals. The system delivers 100 mcg/hr of fentanyl through the skin over a period of 72 hours.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.