Product Images Wixela Inhub
View Photos of Packaging, Labels & Appearance
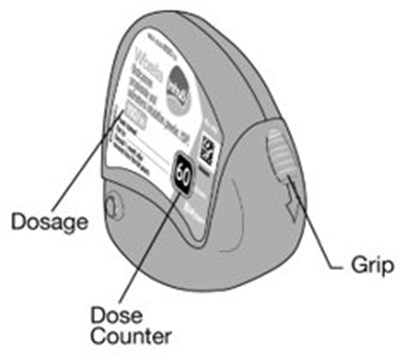
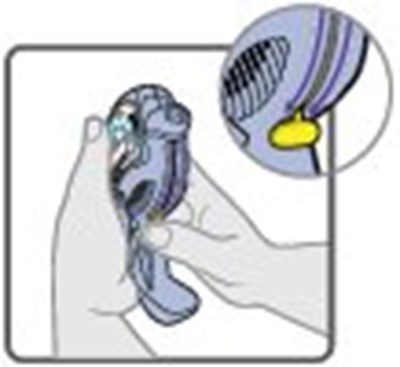
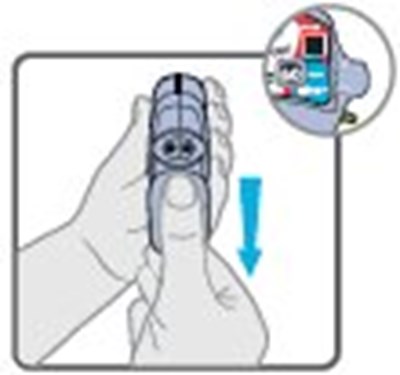
- Instructions for Use Figure A - image 01
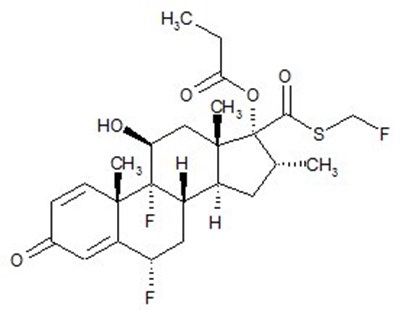
- Fluticasone Propionate Structural Formula - image 02
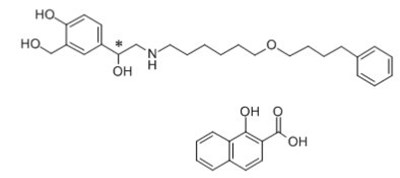
- Salmeterol Zinofoate Structural Formula - image 03
- Figure 1. Mean Percent Change from Baseline in FEV1 in Subjects with Asthma Previously Treated with Either Inhaled Corticosteroids or Salmeterol (Trial 1) - image 04
- Figure 2. Percent Change in Serial 12-Hour FEV1 in Subjects with Asthma Previously Using Either Inhaled Corticosteroids or Salmeterol (Trial 1) - image 05
- Figure 3. Percent Change in Serial 12-Hour FEV1 in Subjects with Asthma Previously Using Either Inhaled Corticosteroids or Salmeterol (Trial 1) - image 06
- Figure 4. Predose FEV1: Mean Percent Change from Baseline in Subjects with Chronic Obstructive Pulmonary Disease - image 07
- Figure 5. Two-Hour Postdose FEV1: Mean Percent Changes from Baseline over Time in Subjects with Chronic Obstructive Pulmonary Disease - image 08
- Instructions for Use Figure B - image 09
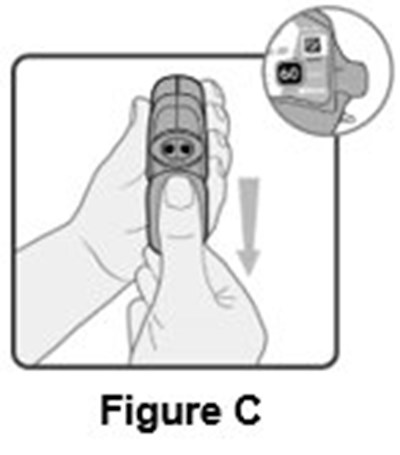
- Instructions for Use Figure C - image 10
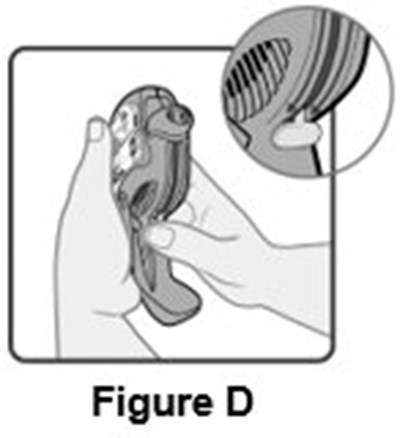
- Instructions for Use Figure D - image 11
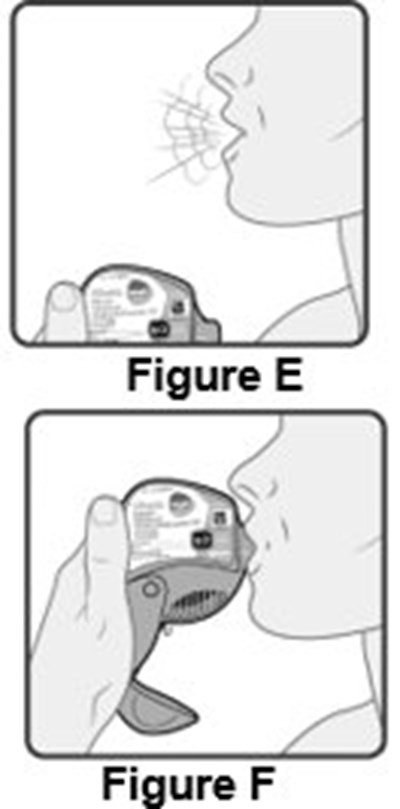
- Instructions for Use Figure F - image 12
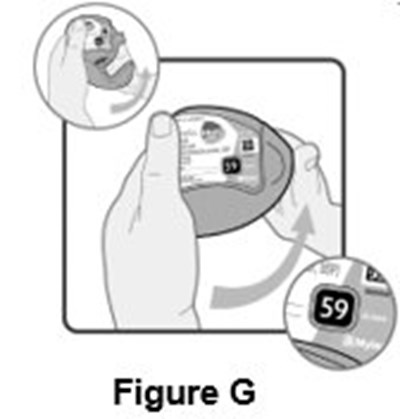
- Instructions for Use Figure G - image 13
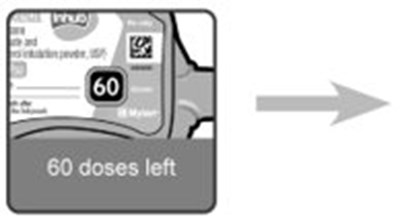
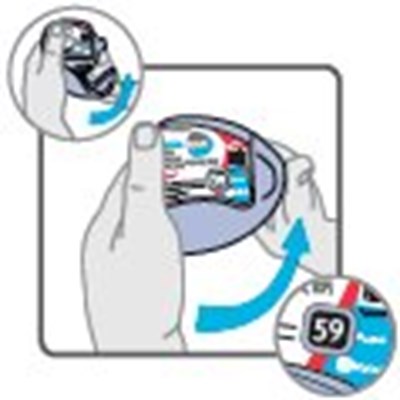
- Instructions for Use Dose Counter Figure 1 - image 14
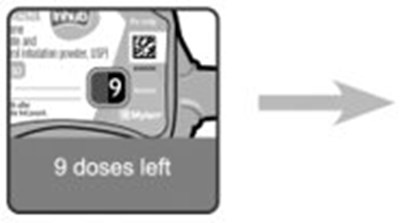
- Instructions for Use Dose Counter Figure 2 - image 15
- Instructions for Use Dose Counter Figure 3 - image 16
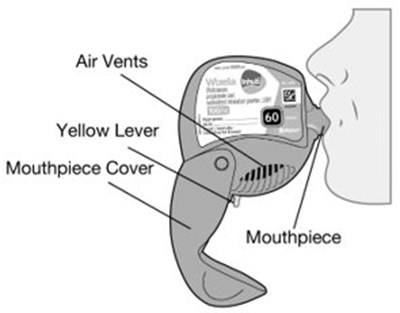
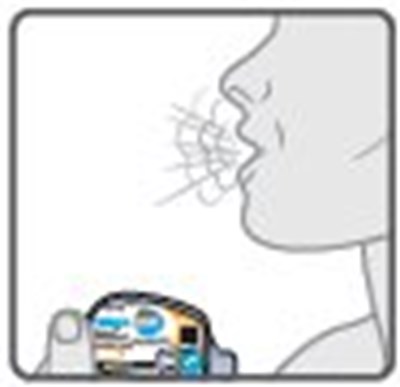
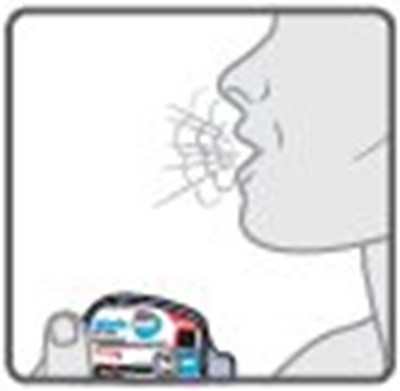
- Instructions for Use Figure E - image 17
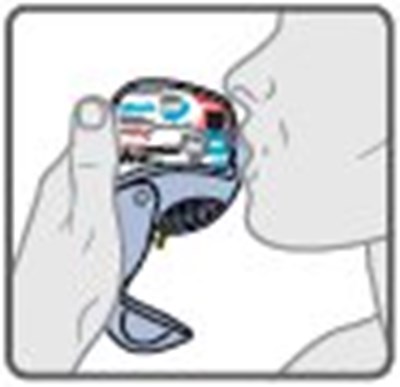
- Instructions for Use Figure H - image 18
- Wixela 100/50 Carton Label Step 3a - image 19
- Wixela 100/50 Carton Label Step 2 - image 20
- Wixela 100/50 Carton Label Step 3b - image 21
- Wixela 100/50 Carton Label Step 1 - image 22
- Wixela 100/50 Carton Label Step 4 - image 23
- Wixela 250/50 Carton Label Step 3a - image 24
- Wixela Inhalation Powder 100/50 Carton Label - image 25
- Wixela 250/50 Carton Label Step 3b - image 26
- Wixela 250/50 Carton Label Step 1 - image 27
- Wixela 250/50 Carton Label Step 2 - image 28
- Wixela Inhalation Powder 250/50 Carton Label - image 29
- Wixela 250/50 Carton Label Step 4 - image 30
- Wixela 500/50 Carton Label Step 3a - image 31
- Wixela 500/50 Carton Label Step 1 - image 32
- Wixela 500/50 Carton Label Step 2 - image 33
- Wixela 500/50 Carton Label Step 3b - image 34
- Wixela 500/50 Carton Label Step 4 - image 35
Product Label Images
The following 35 images provide visual information about the product associated with Wixela Inhub NDC 0378-9320 by Mylan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. Mean Percent Change from Baseline in FEV1 in Subjects with Asthma Previously Treated with Either Inhaled Corticosteroids or Salmeterol (Trial 1) - image 04

This appears to be a table of data related to the use of Fluicasone propionate and Salmeterol as a treatment for some medical condition. There are some numerical values mentioned such as the quantity of Fluicasone propionate and Salmeterol to be taken twice daily. The table also seems to indicate the progress of the treatment over several weeks. However, without additional context or better results, it is difficult to provide further details.*
Figure 2. Percent Change in Serial 12-Hour FEV1 in Subjects with Asthma Previously Using Either Inhaled Corticosteroids or Salmeterol (Trial 1) - image 05

The text describes a table or graph showing the percentage change in FEV on the first treatment day of four different medications, including Fluticasone propionate and Salmeterol. The number of participants for each medication is also mentioned. A baseline measurement is also shown.*
Figure 3. Percent Change in Serial 12-Hour FEV1 in Subjects with Asthma Previously Using Either Inhaled Corticosteroids or Salmeterol (Trial 1) - image 06

Figure 4. Predose FEV1: Mean Percent Change from Baseline in Subjects with Chronic Obstructive Pulmonary Disease - image 07

This is a description of a study that evaluates the efficacy of Fluticasone propionate and Salmeterol inhalation powder in patients with a baseline forced expiratory volume (FEV) of 1207 mL. The patients were divided into two groups, one group received 250mog/50mog of Fluticasone propionate and Salmeterol twice daily, and the other group received a placebo. The study reports the percentage change in FEV at various endpoints.*
Figure 5. Two-Hour Postdose FEV1: Mean Percent Changes from Baseline over Time in Subjects with Chronic Obstructive Pulmonary Disease - image 08

This appears to be a medical study's results regarding the impact of using Fluticasone propionate and Salmeterol inhalation to improve FEV (forced expiratory volume) in individuals. The study involved using different doses of Fluticasone propionate and a placebo. The results show the changes in FEV after different treatments.*
Wixela 100/50 Carton Label Step 4 - image 23

Wixela is a medication used for oral inhalation only. It contains fluticasone propionate and salmeterol inhalation powder. The dosage should be taken as directed by your doctor. The medication comes in a powder form, which needs to be inhaled. The inhaler needs to be stored at a controlled temperature between 20°C and 25°C. The inhaler needs to be discarded after use. After inhalation, you should rinse your mouth with water. If you face any issues or have any doubts, consult your doctor.*
Wixela 500/50 Carton Label Step 4 - image 35

Wixela is a prescribed inhalation powder, for oral use only. It contains fluticasone propionate and salmeterol, which are bronchodilators that help to make breathing easier by relaxing muscles in the airways. Each container holds 60 doses of inhalation powder. Before using it, read the enclosed patient information. The inhaler is easy to use, and each dose contains 500 mcg of fluticasone proponats and 72.5 g of salmeterol base. After use, rinse your mouth with water. Store the product in a cool, dry place, between 68-77 degrees Fahrenheit.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.