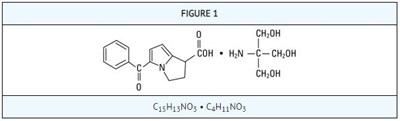
Product Images Ketorolac Tromethamine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Ketorolac Tromethamine NDC 0404-9883 by Henry Schein, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image1.jpg - Image1

The table provides the approximate average pharmacokinetic parameters for Ketorolac Trometharmine. It shows values for oral, intramuscular, and intravenous doses, including bioavailability, time-to-peak plasma concentration, % dose metabolized, % dose excreted in urine/feces, plasma protein binding, and volume of distribution. The values for single-dose and steady-state are also specified, alongside simulated data.*
Image2.jpg - Image2

This is a table showing the influence of age, liver and kidney function on the clearance and half-life of Ketorolac Tromethamine in adult populations. The data is organized according to type of subjects and the type of administration (oral or intramuscular). The table shows the total clearance and the terminal half-life in liters/hours/kilogram. The table provides means and ranges for each subject group. The data is estimated from single doses of Ketorolac Tromethamine.*
Image3.jpg - Image3

This is a list of possible side effects associated with a medication or a medical condition. The side effects include gastrointestinal issues such as abdominal pain, constipation, diarrhea, dyspepsia, flatulence, fullness, ulcers, gross bleeding/perforation, heartburn, nausea, stomatitis, and vomiting. Other possible experiences include abnormal renal function, anemia, dizziness, drowsiness, edema, elevated liver enzymes, headaches, hypertension, increased bleeding time, injection site pain, pruritus, purpura, rashes, tinnitus, and sweating. These side effects have an incidence greater than 10%.*
Image4.jpg - Image4

This is a table showing the incidence of serious gastrointestinal bleeding in patients treated with Ketorolac Tromethamine Injection up to 5 days, based on age, total daily dose, and history of G.I. problems. The data is divided into two sections: one for adult patients with no history of G.I. bleeding, and the other for adult patients with a history of it. The table shows the incidence of bleeding for each category based on the patient's age and the total daily dose of the medication.*
Image5.jpg - Image5

The NDC Number for Ketorolac Tromethamine Injection USP and its package factor is available. The product is available in three variations with different doses in each single-dose vial. The product is packed in 25 vials per carton. The highest dose product is specified for intramuscular use only.*
Label1.jpg - Label1

Description not available. The text appears to be a list of product information including a medication name, lot number, and prescription details.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.