Product Images Midazolam
View Photos of Packaging, Labels & Appearance
- Formula1.jpg - Formula1
- Formula2.jpg - Formula2
- Image1.jpg - Image1
- Image10.jpg - Image10
- Image11.jpg - Image11
- Image12.jpg - Image12
- Image13.jpg - Image13
- Image14.jpg - Image14
- Image15.jpg - Image15
- Image2.jpg - Image2
- Image3.jpg - Image3
- Image4.jpg - Image4
- Image5.jpg - Image5
- Image6.jpg - Image6
- Image7.jpg - Image7
- Image8.jpg - Image8
- Image9.jpg - Image9
- Label1.jpg - Label1
Product Label Images
The following 18 images provide visual information about the product associated with Midazolam NDC 0404-9912 by Henry Schein, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image10.jpg - Image10

This text provides instructions for intramuscular administration of midazolam for sedation/anxiolysis/amnesia prior to anesthesia or procedures in pediatric patients. It advises that higher doses may result in deeper and more prolonged sedation and gives an effective dose range of 0.1 to 0.15 mg/kg with a maximum total dose of usually no more than 10 mg. If midazolam is given with an opioid, the initial dose of each must be reduced.*
Image11.jpg - Image11
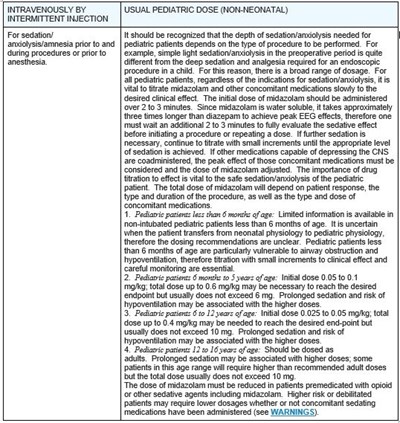
This text provides instructions about the dosage of midazolam for pediatric patients for sedation, analgesia or amnesia before surgery or procedures requiring sedation/anxiety relief. The text warns about the importance of individualization of the dose and monitoring of the patient's response. The dosage is provided for pediatric patients aged less than 6 months, from 6 months to 3 years, from 8-12 years and 12 years and above. Concomitant administration of drugs that suppress the central nervous system must also be taken into account while designing the course of treatment.*
Image12.jpg - Image12

This text provides instructions regarding the dosages of midazolam for pediatric patients in critical care settings. A loading dose of 0.05 to 0.2 mg/kg is given intravenously over 2 to 3 minutes to initiate sedation/anxiolysis/amnesia depending on the clinical effect being aimed for. However, it should not be administered as a rapid intravenous dose. A continuous intravenous infusion may also be given to maintain the effect. The rate of infusion is generally between 0.06 to 0.12 ma/kg/hr and can be adjusted as required or supplemental intravenous doses of midazolam can be given to achieve the desired effect. It is crucial to monitor the patients’ vital parameters and observe for adverse effects. Patients with liver dysfunction, low cardiac output, and neonates may experience delayed drug elimination. Additionally, hypotension may be observed in patients receiving opioids and where midazolam is rapidly administered.*
Image13.jpg - Image13

This text provides information on the continuous intravenous infusion of midazolam for sedation in critical care settings for neonates whose trachea was intubated. It gives guidelines on the usual neonatal dose based on pharmacokinetic parameters and clinical experience, which vary depending on the age of the neonate. Loading doses are not recommended, and infusion rates should be carefully assessed and adjusted to avoid drug accumulation and potential adverse effects. The text also cautions about the risk of hypotension and apnea in critically ill and preterm patients, especially those receiving fentanyl or whose trachea is not intubated.*
Image14.jpg - Image14

Product No. 410102 has an NDC No. of 63323-411-12 and comes in a 2mL vial with a strength of 2mg per 2mL (1 mg per mL). Product No. 411105 has an NDC No. of 63323-411-25 and comes in a 5mL vial with a strength of Smg per SmL (1 mg per mL). Product No. 410110 has an NDC No. of 63323-411-10 and comes in 2 10mL vials with a fill volume of 10mL and a strength of 10 mgper 10mL (1 mg per ml).*
Image2.jpg - Image2
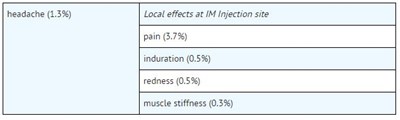
This text appears to be a list of potential side effects and their frequency percentages for a medication that is likely administered via injection. The listed side effects include headache at 1.3%, pain at 3.7%, induration at 0.5%, redness at 0.5%, and muscle stiffness at 0.3%. Additionally, there is a note about local effects at the injection site.*
Image3.jpg - Image3

This appears to be a list of possible side effects, with the percentage of occurrence noted in parentheses, for a medication or injection administered at a particular site. Some of the mentioned side effects include hiccoughs, nausea, vomiting, tenderness, pain during injection, coughing, redness, oversedation, induration, headache, phlebitis, and drowsiness.*
Image4.jpg - Image4

This is a set of guidelines for using midazolam as a preoperative sedative/anxiolytic. The drug should be administered intramuscularly deep in a large muscle mass about an hour before surgery. The recommended dose for good risk adult patients under 60 years old is 0.07-0.08mg/kg. The dose must be reduced for higher risk patients, older than 60 years, and patients who have received concomitant narcotics or other CNS depressants. It also warns that patients must be observed for signs of cardiorespiratory depression after receiving IM midazolam. Onset time is within 15 minutes, peaking at 30 to 60 minutes. Concomitant administration with atropine sulfate or scopolamine hydrochloride and reduced doses of narcotics is possible.*
Image6.jpg - Image6

Continuous infusion of midazolam is a sedation technique that involves the gradual administration of midazolam diluted to 0.5 mg/mL with either 0.9% sodium chloride or 5% dextrose in water. The usual adult dose is a slow infusion of 0.02 to 0.10 mg/kg/hr for maintenance of sedation. Midazolam is a variable drug; thus, the infusion rate should be titrated to the desired level of sedation, taking into account the patient's clinical status, age, and medications. Regular assessments of sedation should be done, and the infusion rate adjusted by 25% to 50% if necessary. Administering opioids reduces the minimum effective midazolam infusion rate.*
Image7.jpg - Image7

The text describes the administration of midazolam in pediatric patients based on their weight. Children usually require higher doses of midazolam (mg/kg) compared to adults, and younger children may require closer monitoring. Obese children's doses should be calculated based on their ideal body weight. The use of opioids or sedatives in combination with midazolam can increase the risk of respiratory depression, airway obstruction, and hypoventilation. The healthcare practitioner should follow the accepted professional guidelines for pediatric sedation appropriate to their situation when using this medication in pediatric patients.*
Image8.jpg - Image8

The text is describing an Observer's Assessment of Alertness Sedation using assessment categories such as Responsiveness, Speech, Facial Expression, and a Composite Score. The scores range from 1 to 5, with 5 being the highest level of alertness and responsiveness and 1 indicating a deep sleep with no response to mild prodding or shaking.*
Image9.jpg - Image9

This is a table presenting the frequency of an observer's assessment of sedation composite scores of pediatric patients undergoing procedures with intravenous midazolam for sedation. The table presents the age range in years and the corresponding number of patients with specific sedation scores ranging from deep sleep to alert.*
Label1.jpg - Label1

This is a description of a drug called Midazolam. It is available in a vial of 10 ml with a concentration of 1 mg per ml. It can only be used for intravenous or intramuscular injection, and it contains benzyl alcohol as a preservative. The container closure is not made of natural rubber latex. Each milliliter of the drug contains Midazolam hydrochloride equivalent to 1 mg Midazolam, and it is combined with 0.3% sodium chloride, 0.01% Edetate disodium, and 100mg of benzyl alcohol. The pH is adjusted to 3.6 with hydrochloric acid. The drug should be kept out of the reach of children and stored at controlled room temperature. The description also includes a lot number and manufacturer details.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




