Product Images Midazolam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Midazolam NDC 0404-9913 by Henry Schein, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Formula2.jpg - Formula2
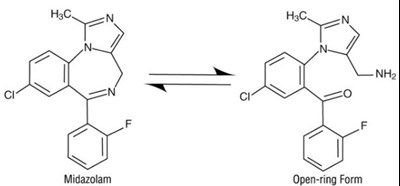
Midazolam Open-ring Form is a type of medication that is used as a sedative and anesthetic. It is commonly used before medical procedures, such as surgery, to help relax patients and reduce anxiety. Midazolam works by binding to specific receptors in the brain that produce a calming effect. The open-ring form of Midazolam refers to a structural change in the molecule that makes it more water soluble and easier to administer into the bloodstream. This form of the medication is commonly used in hospital settings and has been shown to be safe and effective when used appropriately under medical supervision.*
Image2.jpg - Image2

Possible description: This is a list of reported symptoms and their frequencies. Headache occurred in 1.5% of cases and there is a word that was not able to recognize, possibly related to eosinophilia or erythema. Pain was reported in 3.7% of cases. Ndura...ton, which is not a recognizable medical term, and redness each occurred at a frequency of 0.5%. Muscle stiffness was reported in 0.3% of cases. The context and source of this data are not provided, so it is difficult to infer more information.*
Image3.jpg - Image3

This appears to be a list of possible side effects of a medication, including hiccoughs, nausea, vomiting, coughing, oversedation, headache, and drowsiness. It also mentions local effects at the injection site, such as tenderness, pain, redness, induration, and phlebitis.*
Image7.jpg - Image7

The text describes the various strengths and packaging sizes of midazolam hydrochloride in vials. The concentration of the drug is either 1 mg/mL or 5 mg/mL, and the vials come in sizes ranging from 1 mL to 10 mL. The NDC codes for each packaging size are also provided.*
Label1.jpg - Label1

This is a product description for Midazolam Hydrochloride injection, for IM or IV use only, that contains benzyl alcohol. Each milliliter contains Midazolam Hydrochloride, equivalent to 1mg of Midazolam, sodium chloride, edetate disodium, and benzyl alcohol as a preservative. The pH is adjusted to 3.5-3.7 with sodium hydroxide and, if necessary, hydrochloric acid. The product should be kept out of children’s reach and stored at a temperature of 20°C to 27°C (68°F to 77°F). The item number is 24B0885 and the lot number is XXXXXOOXKX with an expiration date in mm-yy format. The product is packaged by Henry Schein, Inc. The GTIN number is (01)20000000000000C and SER number is (21)000000000000C. Additional information can be found in the product's prescribing information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




