Product Images Quetiapine Fumarate Extended-release
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Quetiapine Fumarate Extended-release NDC 0406-1183 by Specgx Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
que150mg60tablets - que150mg60tablets

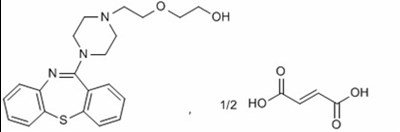
This is a medication guide for Quetiapine Fumarate, an extended-release tablet, which is only available through a prescription. Each tablet contains 173mg of Quetiapine Fumarate and 1509mg of Getapre. The medication should be stored below 25°C and excursions from 15-30°C are permitted. The usual dosage is one tablet alongside the prescription of a doctor.*
que200mg60tablets - que200mg60tablets

This is a medication guide for Quetiapine Fumarate, with each tablet containing 230mg of Quetiapine. The tablets are extended-release and should be swallowed whole once daily. The usual dosage, along with warnings and storage instructions, are included. The manufacturer is Intellipharmaceutics Corp, and the distributor is SpecGx LLC, with a reference to Mallinckrodt Pharmaceuticals.*
que300mg60tablets - que300mg60tablets

This is a medication guide for Quetiapine Fumarate Extended-Release Tablets USP. The tablets come in 345 mg dose and are equivalent to 300 mg of quetiapine. The guide includes instructions to swallow the tablets whole and not to crush or chew them. The usual dosage is once daily, as per the accompanying prescribing information. The guide includes a warning to keep the medication out of the reach of children, and to store it at controlled room temperature (25°C/77°F) with excursions permitted up to 15-30°C (59-86°F). The tablets are manufactured by Intelipharmaceutics Corp. and distributed by SpecGx LLC, and the medication is available through prescription only by Mallinckrodt Pharmaceuticals.*
que50mg60tablets - que50mg60tablets

This is a medication called "quetiapine fumarate" that comes in a bottle with 60 tablets each containing 56 mg of the medication. Patients must be given a medication guide with the prescription and swallow the tablets whole. The usual dosage is once daily, as directed in the prescribing information. The medication must be stored at a temperature of 31-25°C (1°F), with excursions permitted up to 15-30°C (52.6°F). This medication is manufactured by Inelighamaceutcs Co. SpecGxLLC.*
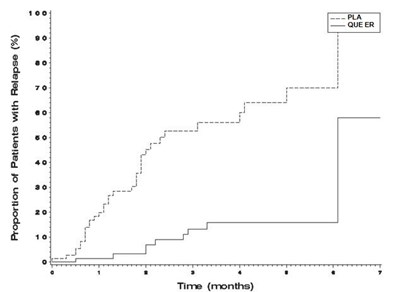
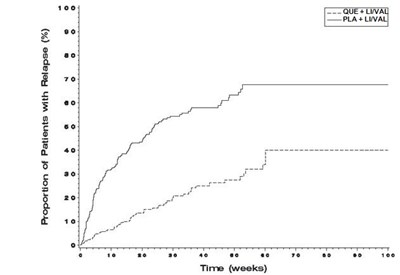
quefig3 - quefig3

This is a graph showing the proportion of patients with relapse over time in weeks. The percentage of patients with relapse is consistently at 100%.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.