Product Images Docetaxel
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 19 images provide visual information about the product associated with Docetaxel NDC 0409-0368 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - docetaxel 02

The text describes a figure labeled as "Figure 1-TAX316 Disease Free Survival K-M Curve". It appears to be a graph showing the disease-free survival rates over time (in months) with percentages indicated on the Y-axis and time indicated on the X-axis. However, the graph seems to be corrupted with random symbols and characters, making it difficult to interpret.*
Figure 2 - docetaxel 03

This is a description of a figure labeled as "Figure 2 - TAX316 Overall Survival K-M Curve". The graph shows the overall survival of a group of individuals over time, with the x-axis displaying survival time in months and the y-axis displaying the probability of survival. Specific time intervals of 6 months are marked on the x-axis up to 66 months. No further information is available to provide additional context or interpretation of this graph.*
Figure 3 - docetaxel 04

The text describes a chart showing the survival probability over time for two different treatments for TAX317. One treatment involves Docetaxel at a dose of 75 mg/m?, while the other treatment involves Best Supportive Care. The chart plots survival probability on the y-axis and time in months on the x-axis. The chart shows cumulative probabilities for both treatments at various time points up to 15 months.*
Figure 4 - docetaxel 05
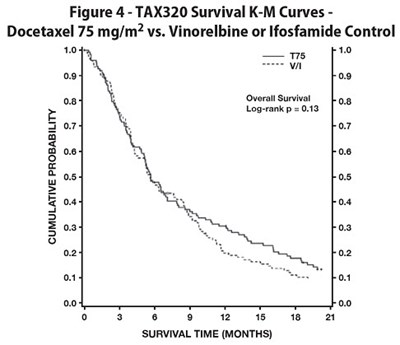
The image shows a graph with survival curves for TAX320. The comparison is between Docetaxel 75 mg/m? and Vinorelbine or Ifosfamide control. The graph shows the survival time (in months) on the X-axis and the percentage of survival on the Y-axis. The graph has Logrankp=0.13.*
Figure 5 - docetaxel 06

The text is describing a graph with survival probability represented by the K-M curves. The graph shows the survival probability over time, in months. However, there is no information on what is being measured for survival probability.*
Figure 6 - docetaxel 07
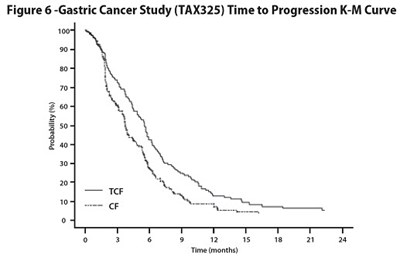
The text represents a figure titled "Gastric Cancer Study (TAX325) Time to Progression K-M Curve". The figure contains a graph showing the probability of time to progression for gastric cancer patients over a period of several months. Unfortunately, due to the lack of context, it is not possible to provide more information on the data and its significance.*
Figure 7 - docetaxel 08

This is a Survival K-M Curve for Gastric Cancer Study (TAX325). It shows the survival rate over time in months. The x-axis shows the survival time in months and the y-axis shows the survival rate in percentage. The curve shows a steady decline in survival rate over time. The data is presented in a graph format.*
Figure 8 - docetaxel 09

The image shows a graph, specifically a Kaplan-Meier curve for the TAX323 study. The x-axis shows the progression-free survival time in months, while the y-axis shows the PFS probability percentage. The curve represents the likelihood of patients in the study remaining progression-free over time. The data points on the curve indicate the probability at specific time intervals.*
PRINCIPAL DISPLAY PANEL - 1 mL Vial Label - docetaxel 13

Docetaxel Injection is a single-dose vial with a concentration of 20 mg/mL and NDC 0409-0366-01. It is ready to be added to an infusion solution for intravenous use. Any unused portion must be discarded. Prior to preparation, the concentration should be checked as per the package insert's instructions. The product is manufactured by Hospira, Inc., located in Lake Forest, IL60045 USA. Other details are not available.*
PRINCIPAL DISPLAY PANEL - 4 mL Vial Label - docetaxel 15

This is a product description for 4 mL multiple-dose vial of Docetaxel Injection, USP, containing 80 mg/4 mL. The vial is sterile and has a National Drug Code (NDC) number of 0409-0367-01. It is ready to be added to an infusion solution for intravenous use only. The user must check the concentration before preparing the solution. The package insert, with complete instructions, is available for the 20 mg/mL concentration. The lot number for this product is ZHL190071.*
PRINCIPAL DISPLAY PANEL - 4 mL Vial Carton - docetaxel 16

This is a description of a medication, specifically a multiple-dose vial of Docetaxel Injection, with a concentration of 80 mg/4 mL (20 mg/mL). It is ready to be added to an infusion solution for administration. The medication contains Dactol and citric acid monohydrate and is stored between 2°C and 25°C. The medication is a cytotoxic agent and should be handled with caution. The package insert contains complete instructions and dosage information. The medication is distributed by Hospira, Inc.*
PRINCIPAL DISPLAY PANEL - 8 mL Vial Label - docetaxel 17

This is a description of a medication named "Docetaxel Injection". It comes in a multiple-dose vial, with a concentration of 20mg/mL and a total volume of 8mL. The medication is sterile and its NDC number is 0409-0368-01.*
PRINCIPAL DISPLAY PANEL - 8 mL Vial Carton - docetaxel 18

This is a product description for an injection solution called Docetaxel in the Hospira product line. It is a 160mg/8mL (20mg/mL) ready to add to infusion solution. This description includes warnings to keep it away from children, directions to retain in original package, and proper storage between 2-8°C (36-46°F). Additionally, there is a cautionary note stating that it is not to be used for agents. Please see the package insert for complete instructions on usage, dosage, and handling.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





