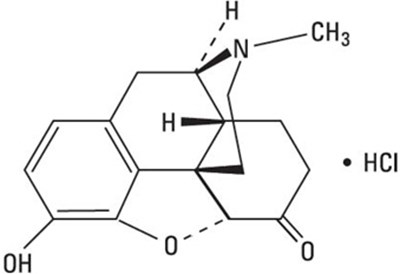
Product Images Hydromorphone Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 18 images provide visual information about the product associated with Hydromorphone Hydrochloride NDC 0409-1312 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure - hydromorphone 03

This is a set of instructions for inspecting a product before use. It advises to check for leaking drug, expiration dates, and particulate matter. The text also cautions against using the product if seals are broken or if it does not match the medication order. There are additional notes advising against placing the syringe in a sterile field and not recapping the needle if utilized.*
Figure - hydromorphone 04
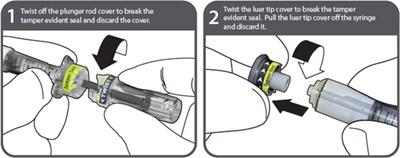
To prepare the syringe, twist off the plunger rod cover to break the tamper evident seal and discard. Then pull the luer tip cover off the syringe and also discard it.*
Figure - hydromorphone 05

This appears to be instructions for administering medication using a syringe. It advises on how to expel air and adjust the dose, as well as connecting the syringe to an appropriate access device based on facility protocol. After administering, the product should be discarded properly.*
PRINCIPAL DISPLAY PANEL - 1 mg/mL Syringe Label - 1283 - hydromorphone 08

This appears to be a label for a single-dose syringe of DROMorphone HCL Injection, USP with lot and expiration date information.*
PRINCIPAL DISPLAY PANEL - 1 mg/mL Syringe Cello Pack Label - hydromorphone 09

This is a description of a medication called Hydrochloride used for injection. It comes in a 1 mL single-dose syringe with a luer lock, sold in packs of 10, and must be prescribed by a doctor. The package insert provides information about the usual dosage. The medication must be stored in a cool, dry place between 20-25°C until it is used, and requires an opaque covering to protect it from light. The text includes product identification and expiration information.*
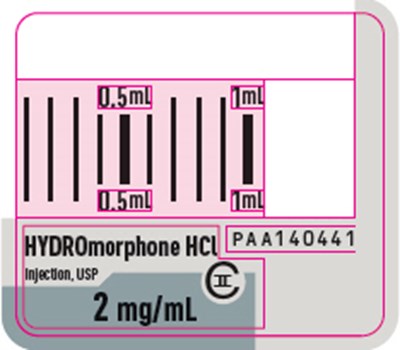
PRINCIPAL DISPLAY PANEL - 2 mg/mL Syringe Label - 1312 - hydromorphone 11

Description: This is a product description of droperidol hydrochloride injection in a single-dose syringe. The syringe has a lot number and an expiry date. The rest of the text contains some coded information related to packaging.*
PRINCIPAL DISPLAY PANEL - 2 mg/mL Syringe Cello Pack Label - hydromorphone 12

This is a description of an injectable medication called HYDROmorphone HCl, which comes in a solution of 2 mg/mL. This medication can be administered through the veins, muscles, or under the skin, and it requires a prescription. The package contains a one milliliter solution with 2mg of HYDROmorphone HCl as well as sodium lactate and sodium chloride to maintain isotonicity. The pH of the solution is adjusted using lactic acid or sodium hydroxide. The product should be stored at a controlled room temperature of 20-25°C, with protection from light until contents are used. The solution is sterile, but if it appears discolored or if it contains a precipitate, it should not be used. The GTIN is 1030409133231, and the expiration date is not available.*
PRINCIPAL DISPLAY PANEL - 4 mg/mL Syringe Label - 1380 - hydromorphone 14

This is a description of a medication called HYDROmorphone HCL provided as an injection in a single dose 1 mL syringe for intravenous, intramuscular, or subcutaneous use. The syringe has a tamper seal, and the medication should be protected from light. The text also includes a code for the medication, prescribing information, and additional details for the packaging.*
PRINCIPAL DISPLAY PANEL - 4 mg/mL Syringe Cello Pack Label - hydromorphone 15

This text describes a medication with the National Drug Code (NDC) 0409-1380-01, which contains hydrochloride as an active ingredient for Intravenous, Intramuscular, or Subcutaneous use. It comes in a package of 10 single-dose syringes with an included luer lock, but without the needle. The medication should be protected from light and stored at a temperature between 20 and 25°C. Additionally, it is notable that the solution should not be used if it is discolored or contains precipitates. The rest of the text appears to be technical details about the product including its ingredient, dosage, and expiration date.*
PRINCIPAL DISPLAY PANEL - 0.5 mg/0.5 mL Syringe Label - hydromorphone 16

This is a label from a medication container. The medication is Droperidol HCL, an injection solution containing 0.5 mg/0.5 mL of the medication. The label also contains information about a "CLEAR-NO COPY" and "OVERLAP AREA," but it is unclear what these terms refer to.*
PRINCIPAL DISPLAY PANEL - 0.5 mg/0.5 mL Syringe Label - 4264 - hydromorphone 17

This appears to be a label or packaging information for a medication. The medication is a solution for injection with a concentration of 0.5 mg/0.5 mL. It can be administered intravenously, intramuscularly, or subcutaneously. The label indicates that the product should be protected from light and is for single use only. There is a tamper seal that can be twisted and pulled to open the package. There are also lot number and expiration date codes present.*
PRINCIPAL DISPLAY PANEL - 0.5 mg/0.5 mL Syringe Cello Pack Label - hydromorphone 18

This is a description for a medication called HYDROmorphone HCI Injection, which contains 0.5mg of hydromorphone hydrochloride per 0.5ml. The injection can be used intravenously, intramuscularly, or subcutaneously and is prescription-only. The package insert should be consulted for appropriate dosage. The medication should be stored in a cool, dry, and dark place until use, and it should not be used if it is discolored or contains a precipitate. The package includes a pharmaceutical identification code and a product code number. The description also includes a lot number and expiration date.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.