Product Images Rocuronium Bromide
View Photos of Packaging, Labels & Appearance
- rocuronium-spl-carton-100-mg - rocuronium spl carton 100 mg
- rocuronium-spl-carton-50-mg - rocuronium spl carton 50 mg
- rocuronium-spl-fig-1 - rocuronium spl fig 1
- rocuronium-spl-fig-2 - rocuronium spl fig 2
- rocuronium-spl-fig-3 - rocuronium spl fig 3
- rocuronium-spl-fig-4 - rocuronium spl fig 4
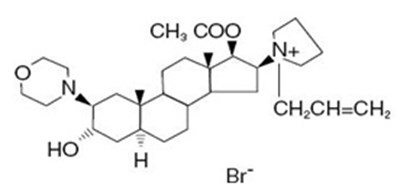
- rocuronium-spl-structure - rocuronium spl structure
- rocuronium-spl-vial-100-mg - rocuronium spl vial 100 mg
- rocuronium-spl-vial-50-mg - rocuronium spl vial 50 mg
Product Label Images
The following 9 images provide visual information about the product associated with Rocuronium Bromide NDC 0409-1403 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
rocuronium-spl-vial-50-mg - rocuronium spl vial 50 mg

NDC 0409-1403-05 is a multiple dose vial of Rocuronium Bromide Injection. Each vial contains 50 mg of Rocuronium in a 5 mL solution. This medication is a paralyzing agent to be used only for intravenous use. It is important to note that it can cause respiratory arrest, so facilities with immediate access to artificial respiration must be available. The medication should be stored in a refrigerator between 2 to 8 degrees Celsius and should not be frozen. Once removed from refrigeration, it should be used within 60 days if stored at room temperature, and opened vials should be used within 30 days. This product is distributed by Hospira, Inc. and the packaging is latex-free.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.