Product Images Butorphanol Tartrate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Butorphanol Tartrate NDC 0409-1623 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - butorphanol 02
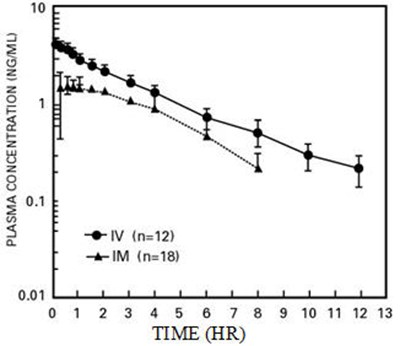
This is a graph with plasma concentration (in ng/mL) on the y-axis and time (in hours) on the x-axis. There are 18 data points in the graph.*
PRINCIPAL DISPLAY PANEL - 2 mg/mL Vial Label - butorphanol 04

This is a prescription drug with the name Butorphanol Tartrate Injection, USP. It is a single-dose medication, and the usual dosage is not mentioned. The drug is recommended for intramuscular or intravenous use, and it is distributed under the name RL-7142 by Hospira, Inc, located in Lake Forest, IL 60045, USA. The National Drug Code (NDC) for this drug is 0409-1626-21. The recommended storage temperature is 20 degrees Celsius.*
PRINCIPAL DISPLAY PANEL - 2 mg/mL Vial Carton - butorphanol 05
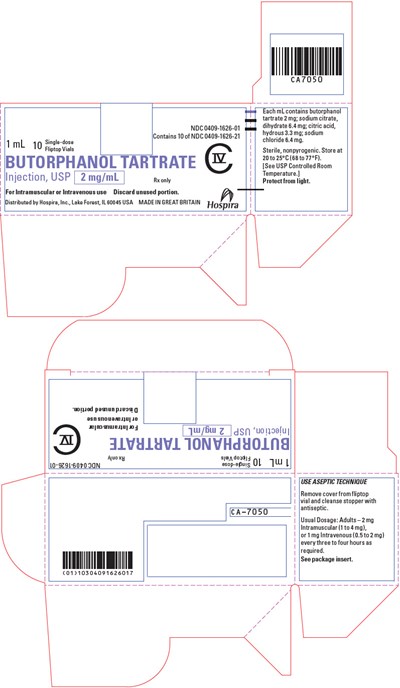
This is a description of a medication called CA7050. It is a single-dose injection that contains 2mg/mL of Butorphanol Tartrate. The medication is intended for intramuscular or intravenous use and is distributed by Hospira. The medication is nonpyrogenic and sterile, and it needs to be stored at a temperature of 20-25°C (68-77°F). Each mL of the medication contains butorphanol tartrate 2mg, sodium citrate dihydrate 6.4 mg, citric acid hydrous 3.3 mg, and sodium chloride 6.4 mg. The dosage for adults is usually 2mg intramuscular or 1 mg intravenous every three to four hours as required.*
PRINCIPAL DISPLAY PANEL - 4 mg/2 mL Vial Label - butorphanol 06

This is a description for an injectable medication called Butorphanol Tartrate. The recommended dosage is not available and the user should refer to the insert. The medication is distributed by Hospira, Inc. and is stored at room temperature. The NDC code for the medication is 0409-1626-42.*
PRINCIPAL DISPLAY PANEL - 4 mg/2 mL Vial Carton - butorphanol 07
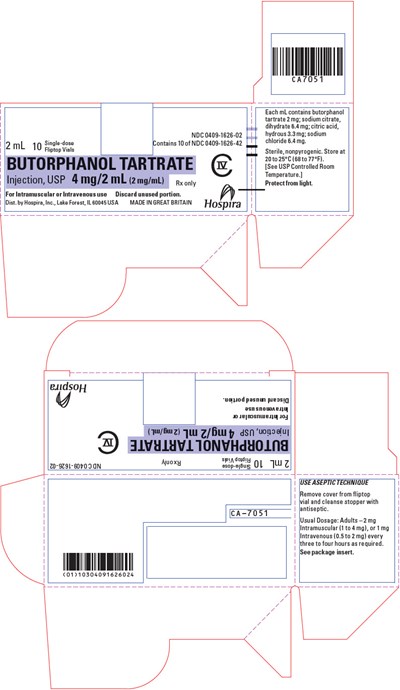
This is a product description for Butorphanol Tartrate injection, a medication used for intravenous or intramuscular use. It comes in a 2ml flip-top vial, with 4mg/2ml strength. Each mL contains Butorphanol tartrate 2mg, and is sodium chloride 6.4 mg. It is sterile and nonpyrogenic, and should be stored at 20-25°C. This medication should be protected from light. This product is manufactured by Hospir, In, and is distributed in the USA and Great Britain. Dosage should be taken as directed by a physician.*
PRINCIPAL DISPLAY PANEL - 1 mg/mL Vial Label - butorphanol 08

This is a description of a medication called BUTORPHANOL TARTRATE in an injectable form with a strength of 1mg/mL. The NDC number is 0409-1623-21, and it should be stored at a temperature of 25°C (77°F). The recommended dosage is available on the insert. It is distributed by Hospira, Inc. in Lake Forest, IL, USA. It is suitable for LM or LV use. The text also contains a random "HiH" at the end.*
PRINCIPAL DISPLAY PANEL - 1 mg/mL Vial Carton - butorphanol 09
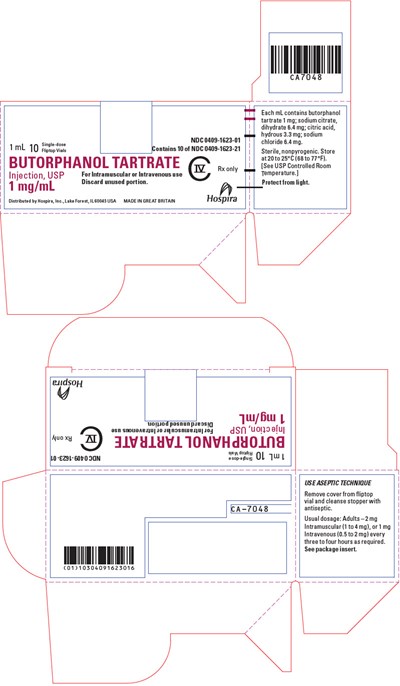
This is a drug label for Butorphanol Tartrate Injection, USP, which contains 1mg/mL Butorphanol Tartrate, and is intended for Intramuscular or Intravenous use. The drug is sterile, non-pyrogenic, and should be discarded if any unused portion is left. The usual dosage for the drug is 2mg Intramuscular, or 1mg Intravenous use for adults, to be repeated every 3 to 4 hours as required. The drug should be protected from light, and should be stored at a temperature of 20 to 25°C. Use aseptic technique while handling the drug.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

