Product Images Hydromorphone Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Hydromorphone Hydrochloride NDC 0409-2634 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Chemical Structure - hydromorphone 01
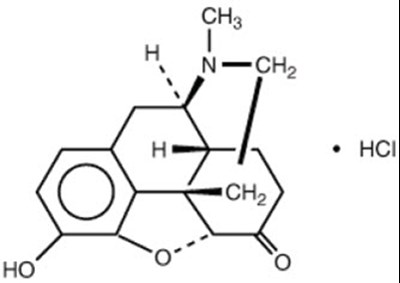
HCI stands for Human-Computer Interaction, referring to the study and design of interactions between people and computers. This field covers a broad range of topics, including user experience design, usability testing, user interface design, and accessibility. HCI research seeks to understand how people interact with technology and how to improve those interactions.*
PRINCIPAL DISPLAY PANEL - 1 mL Vial Label - hydromorphone 03

This is a description of a medication called "Hydromorphone Hydrochloride Injection, USP". The medication comes in a single-dose vial with the National Drug Code (NDC) 0403-2634-10. The medication is a high potency formulation and has a strength of 7.5 milligrams per milliliter. The manufacturer is Hospira, Inc. and it is only available with a prescription. The lot number is indicated as HEHHEAA with an expiration date of DMMMYYYY, but the remaining details are not available due to poor quality.*
PRINCIPAL DISPLAY PANEL - 5 mL Vial Label - hydromorphone 05

This is a description of a high potency formulation of Hydromorphone Hydrochloride Injection, USP. It comes in a 5 mL single-dose vial with an NDC code of 0409-2634-25. It is intended for use in the preparation of 50 mg/5 mL large-volume parenteral solutions, with each mL containing 10 mg of the drug. It is distributed by Hospira, Inc. under the LOT/EXP code of HHEHHEHAA and PAA158974.*
PRINCIPAL DISPLAY PANEL - 5 mL Vial Box - hydromorphone 06
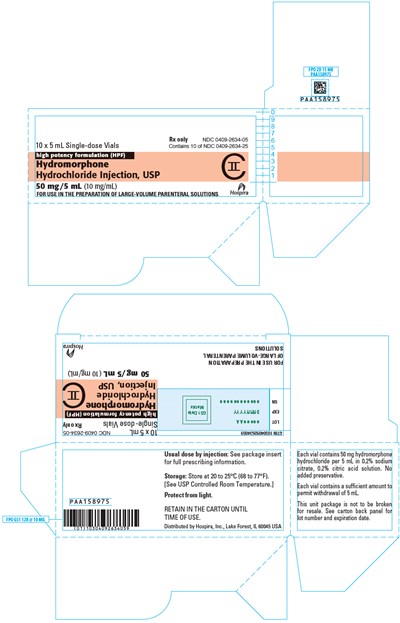
This is a prescription drug that comes in a box with 10 individual 5 mL vials. The drug is Hydromorphone Hydrochloride Injection with a high potency formulation at 50 mg/5 mL (10mg/mL). It is intended for use in the preparation of large-volume parenteral solutions. The drug should be stored at 20 to 25°C (68 to 77°F) and protected from light. The package must not be broken for resale. The carton shows the lot number and expiration date. The usual dose by injection is not mentioned and should be checked through the package insert for full prescribing information. There is no added preservative in each vial.*
PRINCIPAL DISPLAY PANEL - 50 mL Vial Label - hydromorphone 07

This is a description of a single-dose vial of Hydromorphone Hydrochloride Injection, USP in a high potency formulation of 10 mg/mL for use in the preparation of large-volume parenteral solutions. The vial is distributed by Hospira, Inc. and has an NDC of 0409-2634-50.*
PRINCIPAL DISPLAY PANEL - 50 mL Vial Box - hydromorphone 08

This seems to be a mixed output that includes some product labeling information for a medication called Hydromorphone Hydrochloride Injection. It includes details such as the dosage, type of container, and recommended storage conditions. However, some parts of the text are unreadable or contain only symbols, making it difficult to provide a full description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

