Product Images Metoclopramide
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Metoclopramide NDC 0409-3414 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
structural formula metoclopramide hydrochloride - metoclopramide 02
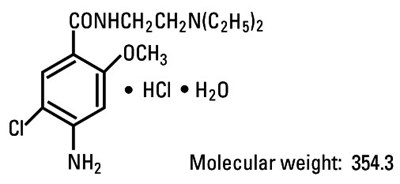
The given text seems to be a chemical formula with molecular weight mentioned as 354.3. Hence, a useful description cannot be generated from this text.*
PRINCIPAL DISPLAY PANEL - 2 mL Vial Label - metoclopramide 05

This is a single-use vial of Metoclopramide Injection, USP with a concentration of 10 mg/2 mL (5 mg/mL) for intravenous or intramuscular use. The vial has an NDC code of 0409-3414-18 and is distributed by Hospira, Inc. The lot number is 08/VP/AP/2013/F/G and the package was approved in August 2013.*
PRINCIPAL DISPLAY PANEL - 2 mL Vial Tray - metoclopramide 06

Each mL of the solution contains metoclopramide 5 mg. Use aseptic technique when handling the solution. Before use, remove the cover from the Flip-top Vial. The solution may contain hydrochloric acid and sodium chloride. Cleanse the top with an antiseptic. The pH level of the solution is approximately 3. Bacterial endotoxins may be present. The remaining text is not readable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


