Product Images Verapamil Hydrochloride
View Photos of Packaging, Labels & Appearance
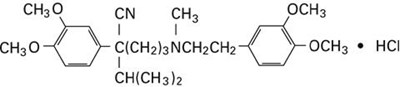
- structural formula verapamil hydrochloride - verapamil 01
- Logo - verapamil 02
- PRINCIPAL DISPLAY PANEL - 4 mL Vial Label - verapamil 03
- PRINCIPAL DISPLAY PANEL - 4 mL Vial Carton - verapamil 04
- PRINCIPAL DISPLAY PANEL - 2 mL Vial Label - verapamil 05
- PRINCIPAL DISPLAY PANEL - 2 mL Vial Carton - verapamil 06
- PRINCIPAL DISPLAY PANEL - 2 mL Ampul Label - verapamil 07
- PRINCIPAL DISPLAY PANEL - 2 mL Ampul Container Label - verapamil 08
- PRINCIPAL DISPLAY PANEL - 4 mL Syringe Label - verapamil 09
- PRINCIPAL DISPLAY PANEL - 4 mL Syringe Carton - verapamil 10
Product Label Images
The following 10 images provide visual information about the product associated with Verapamil Hydrochloride NDC 0409-4011 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 4 mL Vial Label - verapamil 03

This is a description of a medication called AmL Single-dose with the NDC code 0409-1144-62. The medication contains Verapamil HCI and is available in injection form. It should be protected from light and is for L.V. use only. Each dose contains 10 mg of the medication per 4 mL (2.5 mg/mL). The medication is distributed by Hospira, Inc. in Lake Forest, IL, in the United States. The last line contains a barcode.*
PRINCIPAL DISPLAY PANEL - 4 mL Vial Carton - verapamil 04

Verapamil Hydrochloride Injection, USP is a medication that comes in a single-dose Flip-top vial. The vial information include the name of the medicine, the concentration of the drug in the injection, and the protection requirement against light during transportation and storage. The medication is made in Italy and distributed by Hospira, Inc. This drug is intended for intravenous use only and should be used by trained professionals. The text also presents dosage guidelines for Verapamil Hydrochloride Injection, USP.*
PRINCIPAL DISPLAY PANEL - 2 mL Vial Label - verapamil 05

This is a description of a Verapamil HCI injection that comes in a single-dose, with 5mg/2mL, and an NDC code of 0409-1144-65. It is meant for intravenous use only and should be protected from light. The injection is distributed by Hospira, Inc., based in Lake Forest, IL 60045, USA.*
PRINCIPAL DISPLAY PANEL - 2 mL Vial Carton - verapamil 06

Each mL of Verapamil Hydrochloride Inj, USP contains 2.5 mg of verapamil hydrochloride and 8.5 mg of sodium chloride, and may contain hydrochloric acid for pH adjustment. The solution is sterile and nonpyrogenic, with a pH range between 4.0 and 6.5 and an osmolality of 0.301 mOsmol/mL (calc.). It is intended for intravenous use only and should be administered slowly over a period of at least two minutes. Unused portions should be discarded. Dosage information is provided in the insert. The text also contains some unreadable characters, likely due to errors.*
PRINCIPAL DISPLAY PANEL - 2 mL Ampul Label - verapamil 07

This is a description of an injectable medication called Verapamil HCI, which comes in a 5mg/2mL concentration (2.5mg/mL) and is designed for intravenous use only. The medication is packaged with a lot number and expiration date, and should be protected from light.*
PRINCIPAL DISPLAY PANEL - 2 mL Ampul Container Label - verapamil 08

This is a prescription injection medication containing Verapamil Hydrochloride. It is supplied as a 2mL single-dose ampule which should be protected from light and stored at 20-25°C until use. Each ampule contains 2.5mg of Verapamil Hydrochloride and 85mg of sodium chloride. It is intended for intravenous use only and should be injected slowly over a period of at least two minutes. It is recommended to discard any unused portion. The usual dosage should be determined by a medical professional and is available in the insert. This medication is distributed by Hospira, Inc. and is manufactured in Italy.*
PRINCIPAL DISPLAY PANEL - 4 mL Syringe Label - verapamil 09

Verapamil HCI injection is a medication used for intravenous use only. It comes in a single-dose syringe of 4 mL with a concentration of 10 mg/4 mL (2.5 mg/mL). The medication should be protected from light. The manufacturer is Lks Forest and the NDC number for this medication is 0409-9633-65.*
PRINCIPAL DISPLAY PANEL - 4 mL Syringe Carton - verapamil 10

This text contains instructions for using a syringe with aseptic technique, including removing the luer cover and expelling air from the syringe. It also includes details about a medication, Verapamil Hydrochloride Injection, USP, with a concentration of 10 mg/4 mL (2.5 mg/mL). The product is distributed by Hospira, Inc. and made in Italy.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.