Product Images Mannitol
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Mannitol NDC 0409-4031 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 50 mL Vial Label - mannitol 03
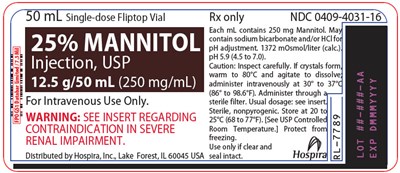
This is a description of a single-dose Fliptop vial available only through prescription, with an NDC code of 0409-4031-16. It contains 250 mg Mannitol per mL and may also contain sodium bicarbonate and/or HCl. The injection must be inspected carefully, should be administered intravenously at a temperature of 30° to 377 (86° to 98.6°F), and must be filtered through a sterile filter. The usual dosage should be seen in the insert. The injection is nonpyrogenic and is intended for intravenous use. It must be stored at room temperature, and protected from freezing, and should only be used if the seal is intact. Severe renal impairment is a contraindication, as mentioned in the insert. The injection is distributed by Hospira, Inc, in Lake Forest, Illinois, USA. However, the last word "Hospr" seems to be not-readable, so it may not be accurate.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


