Product Images Tazicef
View Photos of Packaging, Labels & Appearance
Product Label Images
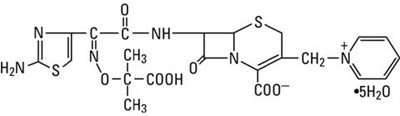
The following 7 images provide visual information about the product associated with Tazicef NDC 0409-5084 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 2 gram Vial Tray Label - tazicef 04

This is a prescription label for Ceftazidime For Injection, USP. The label provides information about the drug such as dosage, composition, adult dose, and route of administration among other details. It also warns about the pressure changes that occur when diluent is added to the vial before reconstitution. Proper storage conditions, packaging, and the manufacturer of the drug are also indicated.*
PRINCIPAL DISPLAY PANEL - 1 gram Vial Tray Label - tazicef 05

This is a description of Ceftazidime Injection, USP, used for infections. The vial should be stored at room temperature and at reduced pressure. The usual adult dosage is 1 gram every 6 to 12 hours or 2 grams every 8 hours for severe infections. Reconstitution instructions and administration are provided in the accompanying prescribing information. Properly reconstituted solutions are stable for 24 hours at room temperature or 7 days if refrigerated. The vial contains ceftazidime pentahydrate, sodium carbonate and sodium at 2 grams and 236 mg respectively. The product is manufactured by Sandoz GmbH for Hospira, Inc. and is sold in single-dose vials.*
tazicef 06

This is the description for a medicine called TAZICEF® which contains 1 gram/vial of ceftazidime. It is used for injection, either intravenously or intramuscularly, and is intended for single-dose use only. The medication comes in a vial that includes ceftazime pentahydrate equivalent to 1 gram cetazidime and 118mg of sodium carbonate. The instructions for reconstitution should be followed carefully. The usual adult dose is 1 gram every 8-10-12 hours. The medication should be protected from light and stored at room temperature between 20°C and 25°C. The manufacturer is Sandoz GmbH for Hospia, Inc, Lake Forest, Illinois, USA.*
tazicef 07

This is a description of TAIZICEF® which is Ceftazidime for Injection, USP, with 25 single-dose vials. It is used for usual adult dosage of 1 gram every 8 to 12 hours. The solution of TAIZICEF is stable for 2 hours at room temperature or 1 day if refrigerated. Each vial contains cetazidime pentahydrate equivalent to 1 gram of ceftazidime and 118 mg of sodium carbonate. The drug is manufactured by Sandoz GmbH for Hospira, Inc. The package should be stored between 2°C to 25°C (8°F to 77°F). The prescription information for reconstitution, dosage, and administration can be found accompanying the product.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.