Product Images Furosemide
View Photos of Packaging, Labels & Appearance
- Hospira logo - furosemide 01
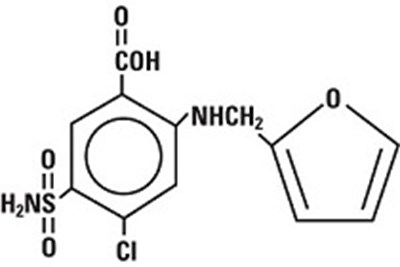
- structural formula furosemide - furosemide 02
- Hospira logo - furosemide 03
- PRINCIPAL DISPLAY PANEL - 10 mL Syringe Label - furosemide 04
- PRINCIPAL DISPLAY PANEL - 10 mL Syringe Carton - furosemide 05
- PRINCIPAL DISPLAY PANEL - 2 mL Vial Label - furosemide 06
- PRINCIPAL DISPLAY PANEL - 2 mL Vial Tray - furosemide 07
- PRINCIPAL DISPLAY PANEL - 4 mL Vial Label - furosemide 08
- PRINCIPAL DISPLAY PANEL - 4 mL Vial Tray - furosemide 09
- PRINCIPAL DISPLAY PANEL - 10 mL Vial Label - furosemide 10
- PRINCIPAL DISPLAY PANEL - 10 mL Vial Tray - furosemide 11
- PRINCIPAL DISPLAY PANEL - 4 mL Syringe Label - furosemide 12
- PRINCIPAL DISPLAY PANEL - 4 mL Syringe Carton - furosemide 13
Product Label Images
The following 13 images provide visual information about the product associated with Furosemide NDC 0409-6102 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 10 mL Syringe Label - furosemide 04

This is a description for Furosemide Injection, USP. It is a single-dose syringe with a 10mg/mL concentration, containing 100mg/10mL of Furosemide for intravenous or intramuscular use manufactured by Hospira, Inc. It is advised not to use the solution if it is discoloured or contains particulates, and there is no preservative present. The packaging includes information about the lot number and expiration date, as well as the contact information of the distributor.*
PRINCIPAL DISPLAY PANEL - 10 mL Syringe Carton - furosemide 05

This is a description of a medication called Furosemide Injection, USP. The concentration of the medication is 100 mg/10 mL (10 mg/mL). It includes instructions for using the medication with the use of aseptic technique. Additionally, the text provides steps on how to prepare the syringe for use. The medication is distributed by Hospira, Inc. in Lake Forest, IL 60045, USA.*
PRINCIPAL DISPLAY PANEL - 2 mL Vial Label - furosemide 06

This is a prescription drug labeled as Furosemide Injection, USP from the manufacturer Hospira with a strength of 20 mg/2 mL (which is equivalent to 10 mg/mL). The National Drug Code (NDC) for this drug is 0409-6102-19. The package contains 2mL of the product and it is distributed in the USA by Hospira, Inc. It is intended for use only under the prescription of a registered healthcare provider. The remaining information seems to be an internal product code or lot number.*
PRINCIPAL DISPLAY PANEL - 4 mL Vial Label - furosemide 08

This is a description for a single-use vial of Furosemide Injection, a medication used for intravenous or intramuscular administration. The vial contains 40 mg/4 mL (10 mg/mL) of the medication, and is distributed by Hospira, Inc. The product code is RL-7531 and the National Drug Code is 0409-6102-18. The medication should not be used if the solution is discolored or contains particulate.*
PRINCIPAL DISPLAY PANEL - 4 mL Vial Tray - furosemide 09

4mL single-use vials of Furosemide injection, USP with 40mg/4mL strength are available with NDC 0409-6102-04 and can be used intravenously or intramuscularly after protecting from light. The vials contain 25 of NDC 0409-6102-18. The rest of the text is not coherent.*
PRINCIPAL DISPLAY PANEL - 10 mL Vial Label - furosemide 10

Description: Furosemide Injection, USP is a medication that comes in a 10 mL single-use vial, for intravenous or intramuscular use. The solution is isotonic and contains furosemide 10 mg and sodium chloride sufficient to render it isotonic. The medication is distributed by Hospira Inc., Lake Forest, IL 60045 USA. However, if the solution is discolored or contains particulate, it must not be used.*
PRINCIPAL DISPLAY PANEL - 10 mL Vial Tray - furosemide 11

This is a description of a medication. The medication is chloride and it is intended for intravenous or intramuscular use. It contains sodium hydroxide and may contain hydrochloric acid for pH adjustment. The solution is sterile and non-pyrogenic and must be sufficient to render the solution isotonic. The usual dosage is not mentioned and must be referred to the insert. It is recommended to not use the solution if it is discolored or contains particulate. The medication should be stored at 20 to 25°C. The medication is manufactured in Italy and distributed by Hospira, Inc, Lake Forest, L 60045 USA. The remaining text appears to be non-sensical and does not provide any information.*
PRINCIPAL DISPLAY PANEL - 4 mL Syringe Label - furosemide 12

This appears to be the label for a medication called "Furosemide Injection, USP" with a concentration of 40 mg/4 mL. It is for prescription use only and does not contain preservatives. The label also includes a National Drug Code (NDC) number and lot number and expiration date information. It is manufactured by Hospira, Inc. There are also some marked spots on the label, but it's unclear what they represent.*
PRINCIPAL DISPLAY PANEL - 4 mL Syringe Carton - furosemide 13

This appears to be a set of instructions for using a medical syringe to administer an injection of Furosemide, a medication used to treat fluid retention in the body, in a sterile manner. The instructions provide guidance on removing the luer cover, relieving any resistance in the barrel, and ensuring that air is expelled from the syringe before injection. The manufacturer is Hospira, Inc., and the product is made in Italy.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.