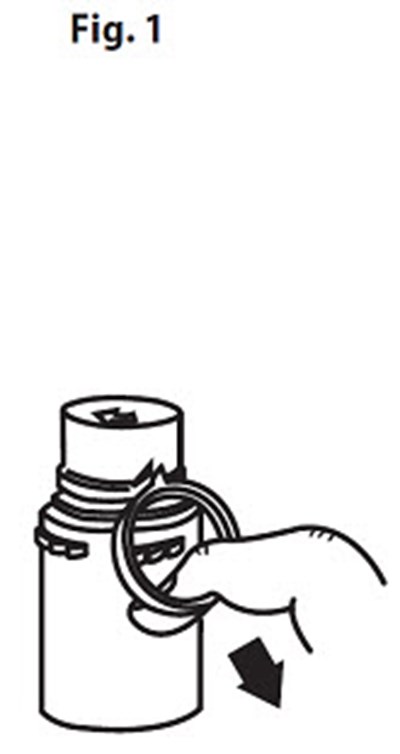
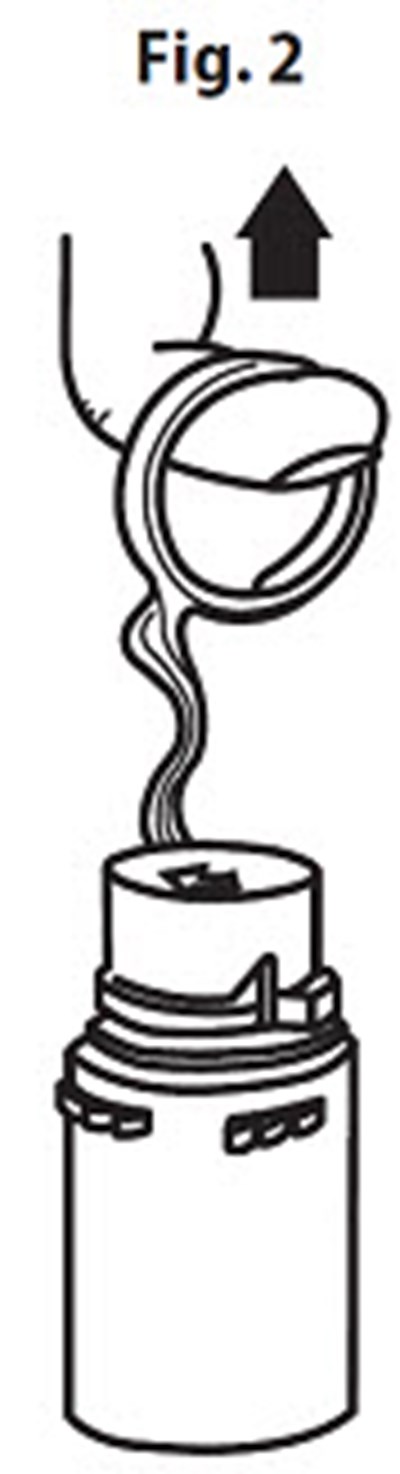
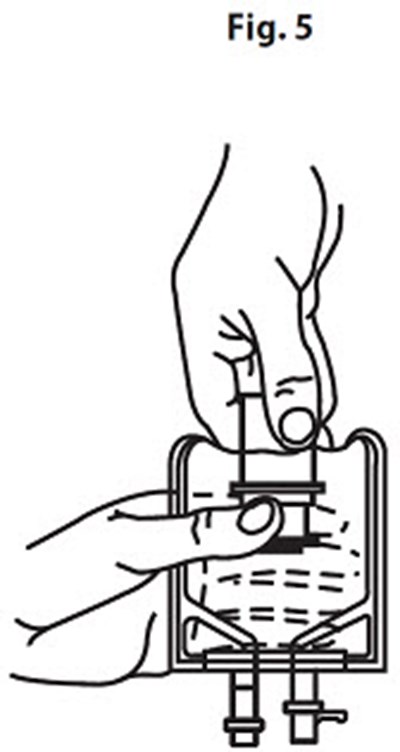
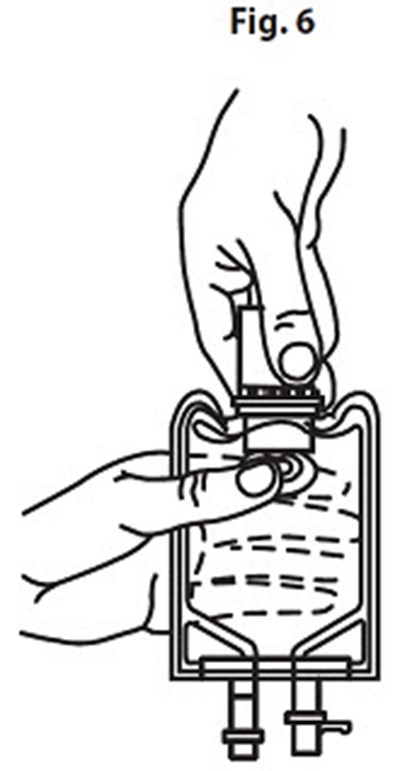
Product Images Vancomycin Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Vancomycin Hydrochloride NDC 0409-6531 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 750 mg Vial Tray - vancomycin 10

This is a description for Singe-Dose ADD-Vansge Viake, which contains Vancomycin Hydrochloride for Injection, USP. The injection is equivalent to 1g/vial and can only be used intravenously, after being diluted as per the instructions provided. Prior to administration, one must read the precautions and directions mentioned on the package. It should be stored between 2°C to 25°C before reconstitution. After reconstitution, it can be stored in a refrigerator for 14 days. The dosage for adults is 2 grams daily in divided doses and the injection is Lyophilized. The product is distributed by Hospira, Inc. in the USA, and might contain Ycrochior acd and/or sodium yiride for PH sdustnent.*
PRINCIPAL DISPLAY PANEL - 1 g Vial Label - vancomycin 11

This is a description for a sterile powder of Vancomycin Hydrochloride for Injection, USP for intravenous use distributed by Hospira, Inc. The vial must be reconstituted and diluted with 250 mL of 0.9% Sodium Chloride Injection or 5% Dextrose Injection. The usual adult dosage is two daily, and it is important to read the insert for precautions and directions before use. The product may contain H and/or NaOH for pH adjustment. FP0 2D 15 Mil and FP0 GS1 DataBar Limited 10 Mil are both types of barcodes on the packaging, with the latter including the barcode number (01)10304096535116. Any other text beyond this is not available.*
PRINCIPAL DISPLAY PANEL - 1 g Vial Tray - vancomycin 12
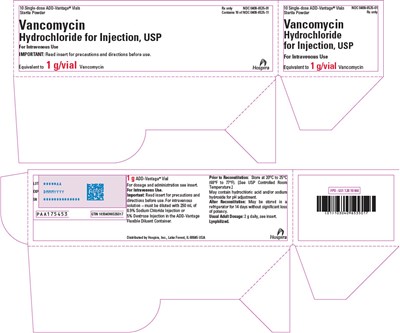
This is a product description for Vancomycin Hydrochloride for Injection, USP. It is used via intravenous injection and comes in the form of a powder in a vial. The vial contains 1g/vial of Vancomycin Hydrochloride. The instructions and cautions are provided on the package, and it must be stored as per the mentioned temperature. The text also mentions the manufacturer's name and address.*
PRINCIPAL DISPLAY PANEL - 500 mg Vial Label - vancomycin 13

This is a product description of a single-dose ADD-Vantage Vial containing sterile powder for injection. The vial has an NDC number of 0409-6534-11 and may contain Hydrochloride and/or NaOH for pH adjustment. The product contains Vancomycin, with an equivalent to 500 mg/vial Lyophilized powder for injection and is distributed by Hospira, Inc. The text seems to be accompanied by two different FPO codes and a lot number (PAA175388), which may pertain to packaging and shipping purposes.*
PRINCIPAL DISPLAY PANEL - 500 mg Vial Tray - vancomycin 14

This is a description of two products of Vancomycin Hydrochloride in powder form for injection purposes. The first product comes in 10 ready vials and the second product comes in 10 single-dose ADD-vantage vials. Both versions contain Vancomycin Hydrochloride equivalent to 500 mg/vial Vancomycin. Further instructions on the insert should be read before use, including room temperature and the practices for reconstitution. The usual adult dose is 2 g daily. The products may need to be adjusted with sodium chloride and/or sodium hydroxide. The brand of the products is Hospira.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.