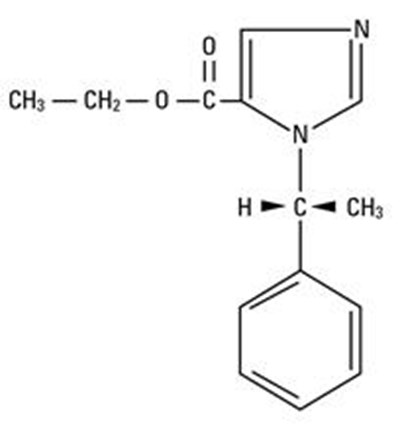
Product Images Amidate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Amidate NDC 0409-6695 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 10 mL Vial Label - amidate 03

This is a description of a drug called Etomidate Injection, USP that comes in a 0 mL single-dose Fliptop vial. Each mL contains 2mg of etomidate and propyleneglycol 35% v/v. It is sterile and monpyrogenic and should only be used intravenously. The product is distributed by Hospira, Inc., based in Lake Forest, IL, USA. There is also a usual dosage that should be referred to before use. The rest of the text is unclear or non-English characters.*
PRINCIPAL DISPLAY PANEL - 20 mL Vial Label - amidate 05

This is a medication for intravenous use only, called Etomidate Injection, which comes in a single-dose Fliptop vial. The NDC code is 0409-6695-12 and it is distributed by Hospira, Inc. Each mL of the injection contains 2 mg of etomidate and propylene glycol. The dosage instructions are included in the insert and any unused portion must be discarded. The text also includes a code, which might be a product code, RL-7186.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.