Product Images Dextrose
View Photos of Packaging, Labels & Appearance
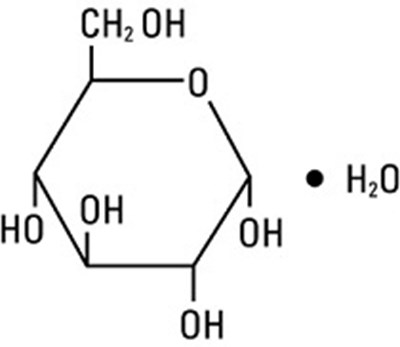
- structural formula dextrose - dextrose 01
- figure 1 - dextrose 02
- figure 2 - dextrose 03
- figure 3 - dextrose 04
- figure 4 - dextrose 05
- figure 5 - dextrose 06
- figure 6 - dextrose 07
- figure 7 - dextrose 08
- Hospira logo - dextrose 09
- Bag NDC 0409-7100-68 - dextrose 10
- Bag NDC 0409-7100-69 - dextrose 11
- PRINCIPAL DISPLAY PANEL - 250 mL Bag Label - dextrose 12
- PRINCIPAL DISPLAY PANEL - Pouch Label - WR-0425 - dextrose 13
- dextrose 14
Product Label Images
The following 14 images provide visual information about the product associated with Dextrose NDC 0409-7100 by Hospira, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Bag NDC 0409-7100-68 - dextrose 10

This is a description of a medical product called ADD-Vantage™ Unit. The text provides directions on how to use the drug, which includes pulling the inner plug/stopper and mixing the drug before use. The product is intended for intravenous use and contains 5% dextrose. The package contains a single-dose container and is sterile and non-pyrogenic. The text warns against the use of dextrose solutions without salts in blood transfusions due to possible rouleau formation. It notes that the product should only be used with ADD-Vantage™ system components and provides the NDC number (0409-7100-68). The text also provides information on the product's pH, osmolality, and dosage information. Finally, there are instructions for ensuring product integrity, including checking for clear solutions and undamaged containers.*
PRINCIPAL DISPLAY PANEL - 250 mL Bag Label - dextrose 12

The text is a warning label on a container of 5% Dextrose Injection for intravenous use with instructions to mix the drug before use and not to use with blood transfusions. It also states that the drug should only be used with ADD-Vantage™ System components and must not be used in series connections. The container is sterile and nonpyrogenic. It is distributed by Hospira, Inc. in Lake Forest, IL, USA. The text contains a lot number, a National Drug Code (NDC) number, and a warning that the solution contains DEHP.*
PRINCIPAL DISPLAY PANEL - Pouch Label - WR-0425 - dextrose 13

This is a description for the One/ADD-Vantage™ unit intended for use with ADD-Vantage™ system components. The overwrap functions as a moisture barrier, and the unit should be used within 30 days of opening the overwrap, provided the use date is before the expiration date. The recommended storage temperature is between 20-25°C (68-77°F), with protection against freezing. After removing the overwrap, the container should be checked for leaks. If any leaks are found, the unit should be discarded as the sterility could be compromised. This product is distributed by Hospira, Inc. from Lake Forest, IL 60045.*
dextrose 14

This text provides instructions for opening and using Five/ADD-Vantage™ Units, which are intended for use with ADD-Vantage™ system components. The overwrap serves as a moisture barrier and units should be used within 30 days of opening, as long as the expiration date is not exceeded. It is important to check for leaks after removing the overwrap; any leaks found may impair sterility and require unit disposal. Proper storage temperature is between 20°C to 25°C. The text also indicates that this product is prescription-only and distributed by Hospira, Inc. in Lake Forest, IL, USA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.